[Ed. Note: Here is the latest from Dr. KSS, who writes about health, medicine and biotech stocks for the Irregulars. He has agreed to our trading restrictions, and his words and ideas are his own.]
Dedicated to Cynthia Chew Choon Hoon (1959-2013). In pace requiescat.
Imagine a day down the hallway of the future. You’re a happy Gumshoe Irregular, you’ve just turned 65, and last week, you had a routine check-up with your doctor. You feel great and are looking forward to a contented, ebullient retirement. It’s early evening. You and your wife are having gin-and-tonics on the veranda, watching the sunset. The lime in your drink is tart, zesty, and life is good.
But the phone rings. Your wife answers, ignoring your importunings that she ignore it. She beckons you. It’s your doctor, and he wants to speak with you. Can’t be good.
The doctor is still in his office going over labs and test results, and decided to call you rather than sending a letter. Last week, he drew a PSA and also examined your prostate. It seemed fine. But it’s your PSA that concerns him. It is now 4.5, not screamingly high, but up from last year’s level of 4. He wants you to come in again.
You imagine a device being shoved into your rectal vault, from which lots of needles will suddenly extrude and plant themselves painfully deep into your prostate gland, translocating germs from your rectum into it, causing bleeding for days on end…and you begin feeling dizzy. Your ears begin to ring, you break out into a cold sweat. Your worst fears are coming true, you begin seeing spots, and slide onto the floor. Little birds fly around your head.
“Hon?! Are you OK?,” your wife shouts.
“Hello……sir? Are you still there?” the doctor is shouting into the phone.
Your wife grabs the phone. “Doctor, my husband just fainted!”
“Is he OK?”
“Just dizzy and sweaty. He’s coming around.”
“I must have scared him. I told him his PSA is high.”
“Yeah, he mumbled something about prostate biopsies? I think that put the fear of God into him.”
“Prostate biopsies? I didn’t say anything about that.”
“Isn’t that what you are planning doctor?,” your wife asks.
“Well, no, not at all. I just wanted him to come back so we could draw blood, you know, to look for any prostate cancer cells. Prostate biopsies? No, we don’t do those anymore.”
Medical science fiction? Probably not. The lab test in question, looking at blood to find circulating tumor cells, is now making its first forays into prime-time, though as of yet it is to assess response to cancer treatment rather than in place of tumor biopsies. But that is likely to change, and in fact one tiny biotechnology company seems uniquely poised to benefit from this new science and this emerging “hot”new diagnostic trend.
Do you remember this scene from The Silence of the Lambs? Have a look. Clarice Starling is wrestling with the identity of a killer, and Hannibal Lecter, trying to act as her mentor, encourages her to think like Marcus Aurelius and mull the essence, the nature, of the killer.
Cancer is a killer. But what is cancer, in its essence? What is the nature of cancer? What distinguishes cancer from all other illnesses? What does cancer do that no other disease does? There’s an old word I love, quiddity, sometimes defined as that which answers the question, quid est? What is the quiddity of cancer?
Cancer spreads. It metastasizes. That is the most scary aspect of its nature. Cancer is like a flea that bounds from place to place in the body, and takes up residence easily at sites remote from where it started. Yes, cancer can extend locally, but it loves to end up at distant sites without leaving any crumbs for how it got there. How does cancer get to those places? Some will answer via the lymphatic system….that cancer goes to lymph nodes and from their more broadly into the body. But there is a better answer.
The fact is, cancer cells get into your bloodstream, and course around as if they were river rafting. And this idea is not a new concept. When they reach a rivulet, a capillary, a tight spot from which they can flow no further, they lodge. They take root there. Most of these attempts at colonizing, at metastasizing, provoke immune reactions that are deadly to the tumor cells.
Armand Trousseau (1801-1867): A Gregarious Master Clinician
Nineteenth-century France gave the world many fine physicians, including Dupuytren, Duroziez, and the astonishing Rene Laennec. But none were more remarkable than Armand Trousseau, an affable Paris internist.
On 27 June 1867, Trousseau died…to the shock of friends, patients, colleagues, seemingly to the surprise of everybody but one person: Armand Trousseau. He was 65. In January of that year, he had noticed something amiss in his left upper arm. One imagines him at a wash basin with a straight razor shaving around his bushy sideburns, and feeling a pang. He noticed a painful, tender, hot mass in his arm, a blood clot. The next day it had subsided and the mass and pain were gone. He knew what was coming, and knew he had at most 6 months.
The eminently likable Armand Trousseau, MD. Il a découvert et puis il l’a tué
Trousseau had many fine accomplishments—better treatments for COPD, malaria, pleuritis, goiter, yellow fever; he performed the first tracheotomy in France—and was also famously known as a patient advocate and a person who became so absorbed in listening to and studying patients at their bedside that he lost track of time. In Irving Stone’s fine historical novel about Sigmund Freud, The Passions of the Mind, he depicts a scene in which, at a teaching hospital bedside, the great Charcot extols the virtues of “see-ing,” of really opening one’s senses and studying patients, of even ignoring one’s education somewhat and letting bedside findings, primary observation, guide you in formulating disease and its treatment. Paracelsus admonished physicians always to let patients be their true textbook, that observing patients never deceives. Trousseau paid attention to patients, and little escaped his notice.
 In Choruses From “The Rock,” T.S. Eliot asks, “Where is the wisdom we have lost in knowledge?” Trousseau would have sympathized with this, because regardless of his learning, regardless of what he had read, he always noticed the details of what he observed with every patient. “Get the patient well!” was what Trousseau always told medical students, and it was with patient centeredness in mind that he noticed a process no one else had: the phenomenon of migratory thrombophlebitis, later known as Trousseau syndrome. Trousseau noticed that certain people would have acute venous thrombosis with local inflammation in a limb, with disappearance of the process one or two days later, and that patients always died within 6 months of a visceral cancer, malignancy somewhere in the abdomen. He hypothesized that cancer somehow distorted the clotting system, favoring thrombosis (he was correct…except that rarely prostate cancer favors a bleeding tendency). Later observers also noticed that lung cancer causes Trousseau syndrome. And in fact, pace Trousseau, patients who have an unexplained episode of deep vein thrombosis need underlying cancer excluded. (Trivia question for medical readers: what is the Trousseau sign?)
In Choruses From “The Rock,” T.S. Eliot asks, “Where is the wisdom we have lost in knowledge?” Trousseau would have sympathized with this, because regardless of his learning, regardless of what he had read, he always noticed the details of what he observed with every patient. “Get the patient well!” was what Trousseau always told medical students, and it was with patient centeredness in mind that he noticed a process no one else had: the phenomenon of migratory thrombophlebitis, later known as Trousseau syndrome. Trousseau noticed that certain people would have acute venous thrombosis with local inflammation in a limb, with disappearance of the process one or two days later, and that patients always died within 6 months of a visceral cancer, malignancy somewhere in the abdomen. He hypothesized that cancer somehow distorted the clotting system, favoring thrombosis (he was correct…except that rarely prostate cancer favors a bleeding tendency). Later observers also noticed that lung cancer causes Trousseau syndrome. And in fact, pace Trousseau, patients who have an unexplained episode of deep vein thrombosis need underlying cancer excluded. (Trivia question for medical readers: what is the Trousseau sign?)
Trousseau was among the earliest popularizers of bedside, patient-based teaching. When he had interesting or complex patients, he would summon students and trainees for long, impromptu sessions in which they gathered around the patient. Trousseau would hold forth, and recite the patient’s history with a novelistic richness, a raconteur’s vim and vigor. This often charmed patients, that he had gone to such pains to know their histories in depth. And he often amazed patients with insights about their lives, their motivations, their relationships….things they recognized as true, but had not recognized before. Many physicians who love teaching at the bedside—and I am one—love it for one reason: the teacher gets to midwife the forging of new neural links, new understanding….gets to be present at the birth of knowledge. Students are asked what they see, and how that may relate to something else they’ve seen. With the right Socratic bent, five minutes at the bedside can teach a student vastly more than six weeks at the books can, but also tends to do so with a suddenness that thrills. The ice floes of book understanding break up, and the white water of real comprehension flows. The student gets a look on his or her face suggesting that serotonin, dopamine, glutamine and norepinephrine are flooding their synapses in unison: they get it now, they never got it before, and they will never forget it. You don’t do this so you can take credit for it, and in fact you do not care if the student even remembers that it was you who helped them learn. You just want to be there for the epiphanies, the fleeting moments when a student’s eyes, brow, and lips convey that they just underwent a cognitive firestorm, and that several brain neurons that had never met are now hopelessly, inextricably intertwined.
At the height of his reputation, Trousseau delivered a fine lecture in Paris on the phenomenon of migratory thrombophlebitis, which he called phlegmasia alba dolens:
“I have long been struck with the frequency with which cancerous patients are affected with painful edema in the superior or inferior extremities, whether or not either was the seat of cancer. This frequent concurrence of phlegmasia alba dolens with an appreciable cancerous tumor led me to the inquiry of whether a relationship of cause and effect did not exist between the two, and whether the phlegmasia was not the consequence of the cancerous cachexia.” (translated from the French)
An autopsy on Trousseau showed he had advanced gastric cancer. The jovial, inspiring doctor was dead. He is still quoted in medical training programs all over this planet for his shrewdest utterance: “utiliser de nouveaux médicaments rapidement, alors qu’ils travaillent encore” (“Use new medicines quickly, while they still work.”) Even before the days of Big Pharma and vaunted claims for new medicines, Trousseau was aware of the meta-placebo effect, placebo by proxy: that sometimes newly-released medicines work, or seem to, simply because physicians have great faith in them.
Theodor Billroth, the famed Prussian-Austrian surgeon who was a contemporary of Trousseau, would go on to identify pancreas cancer cells rarely found embedded with clumps of platelets in the capillaries of pancreas cancer patients. Billroth never published this, but noted it in his personal papers. One wonders if Billroth, who advanced the art of visceral surgery considerably, could have saved Trousseau. Billroth was a personal friend of Johannes Brahms, an excellent musician, and often proofread and helped Brahms rehearse his pieces as he was writing them. Brahms would go on to die of a mysterious jaundicing illness that baffled Billroth, and about which I have long meant to publish a theory.

Theodor Billroth (1829-1894). Before there were Billroth-I and Billroth-II procedures, there was Billroth the man. He performed the first successful gastrectomy for gastric cancer in 1881.
Two years later, in 1869, Thomas R. Ashworth, a Melbourne physician, would publish the literature’s first description of circulating tumor cells in Australian Medical Journal. Ashworth had a patient who died of advanced cancer, and at autopsy found occasional cells in the patient’s blood identical to those in the cancer.
Black swans, hens’ teeth, four-leaf clovers…. and black cats in coal cellars
It’s not clear that Ashworth knew of Trousseau syndrome, or would have appreciated what would later be recognized (vide infra), which is that tumor cells trigger platelet aggregation and that this is responsible for what Trousseau observed. But like many Australians, he was clever: tumors did not metastasize via a Star Trek transporter (or choose a 19th century analogy). Best to go looking for individual tumor cells, then, loose and afoot in the system. He found them….cells in the blood identical to cells in the tumor. Looking for them was like looking for a black cat in a coal cellar. One of the ironies of Ashworth’s description of the patient and the cells is that no one has ever known for sure what kind of cancer the patient had. Ashworth described subcutaneous nodular cancer, and many feel he may have been reporting a case of soft-tissue sarcoma.

White peacocks, red bananas, insanely big crabs. Australia has always given the world rare things, like the discovery of circulating tumor cells. One thinks of black swans as rare, until you cross the Hobart, Tasmania, bridge over a waterway inhabited by them!
When cells grow in organs, they do so in an orderly, non-aggressive, well-behaved way, not violating boundaries, displaying inhibition at contact with neighboring cells. Cancer cells do not scheme badness, do not plot ways to subvert the system. As we are all of us evolved, so cancer represents certain elemental steps that go on in evolution: DNA occasionally rolls the dice, rearranges itself. Occasionally certain rules get subverted. Humans fancy the ideas of immortality like the gods have. And so it, seems, maybe do cells. They stumble upon ways to live forever, and to propagate themselves by growing aggressively and even escaping their confines, becoming flotsam and jetsam that catch blood currents to other locales. It’s not that the cells go rogue and plan to cause trouble. But gene rearrangements and mutations confer properties on cells that harm the organism, and when enough errant properties, including the abilities to invade, metastasize, repel the immune system and resist intrinsic mechanisms of apoptosis come together, the result is cancer and it kills. Humans long for, dream of, immortality. Is there a reason our cells should not do the same? (And is it possible that our craving for immortality is hubristic because it is harmful to some greater system (Jung called it the Collective Unconscious), just as cancer harms us, that we comprise but know only through-glass-darkly?)
If you have cancer, how likely are you to have circulating tumor cells? No one knows. In fact, this is a question today’s featured company may help medicine answer. Certainly the more advanced, the larger, the more inclined to metastatic spread a tumor is, the likelier it is to have breakaway stray cells found in your blood. The number of cells in a cancer is proportionate to the volume of a tumor. From geometry, we know that the volume of a sphere rises as a function of the radius to the third power. Which means that when a tumor doubles its diameter, it has 8 times as many cells as it did at its former size. So the number of circulating tumor cells should be strongly linked to tumor size, and in fact a test for circulating tumor cells as a global screening test for any kind of cancer as part of a wellness check is unlikely to happen. We will always need to continue to pay attention to organs—-breast, prostate, colon, the lungs of smokers, the skin of the Celtic and the immunosuppressed—-known to be trouble spots.
Here are some number to give you a sense of scale for the prevalence of circulating tumor cells. One liter has one million microliters, or 1000 cubic centimeters. One cubic centimeter is one milliliter. Blood has roughly one million red blood cells per microliter. Blood may have about 1000 white blood cells per microliter. But in a cancer patient, blood may have only 100 to 1000 circulating tumor cells per liter; that’s one circulating tumor cell for every one to 10 cc’s of blood.
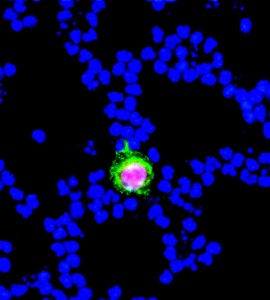
Technology makes it easy to spot this circulating prostate cancer cell in blood. T.R. Ashworth had no technology…it was Australia 1869 for him. (From Genelux.com)
Huahsi Night Market (華西街夜市)
In 1991, I packed and departed for Taipei. Circulating tumor cells have long fascinated me, and I was intrigued by data that circulating tumor cells often break loose only to provoke platelet aggregation around them. The tumor platelet clump may form a bigger vessel for a voyage, and once it lights or lodges at some point, the platelets release molecules that act as growth factors, fertilizer to jazz up tumor growth. I wanted to study this further, and in fact had heard of a viper unique to Taiwan with a venom component that seemed able to halt the process.
Taipei has many fine night markets, but the one at Huashi is notorious. Handlers taunt and irritate cobras while you watch, take extraordinary risks with them, and then kill them, slick them open with a knives or scissors, and empty the snake’s bile and blood into a glass of strong liquor (ask Irregular Eddy to tell you about the vaunted properties of this cocktail). I did not handle any snakes in Taiwan, but we relied on handlers like these to provide the venom for our studies.
We were able to demonstrate that when we grew human breast, prostate or colon cancer cells in culture, and then made a suspension of intact tumor cells, those tumor cells would immediately provoke full aggregation, or clumping together, of platelets. When blood clots, there are two waves of it: primary hemostasis, in which a platelet plug forms at a site of injury, and then secondary hemostasis, in which (as per the article on Regado) insoluble fibrin is formed from soluble proteins in blood. Normal cells do not cause platelets to aggregate. Most tumor cells do, and with considerably potency. What’s more, the avidness with which tumor cells provokes platelets to aggregate, we showed, is proportional to how much metastatic potential they have.
What Trousseau was observing, in fact, was circulating tumor cells (some, but not all) causing tumor-cell-induced platelet aggregation. These clumps would lodge, set off an inflammatory process, but also because not enough molecular stimuli were present to provoke a full and thick clot, would probably set into motion clot-dissolving machinery (fibrinolysis), and the tumor cell/platelet clump break up after 1-2 days.
Tumor cells appear to express uncommonly high levels of tissue factor. As in the Regado article, this is a cofactor for factor VII, and when these two merge, they afford thrombin. Although collagen, epinephrine, ADP and thromboxane are platelet agonists, thrombin is the physiologic platelet agonist, and triggers platelet clumping even when present at concentrations too low to cause fibrin clot formation.
Cancer: Metaphysics, Semiotics, and Metaphors
In the late 90’s, I found myself late one summer in a situation I am sure other readers have. I was exhausted from working hard. I had a vacation week coming, but somehow had planned nothing. But I really needed to do something fun and diversionary. At the very last moment, I booked bargain passage to Budapest, seemingly the last quarter of Europe I’d not seen. I had literally packed for it by throwing things into a knapsack while a cabdriver was outside angrily blowing his horn. I was in a state of physical depletion when I left, so much so that upon renting a car in Budapest, I pulled over in a public park, stretched out on a table, and dozed off and began dreaming. I woke up feeling good, and drove off in earnest toward Slovakia, into a park near the magnificent Aggtelek Cave system, and found a wooden cabin. In chill mountain air, I turned on an incandescent lamp at the head of the bed, reached into the knapsack and fished out two books I had long meant to read: Susan Sontag’s Illness as Metaphor and AIDS and Its Metaphors. I didn’t doze off again til dawn.
Sontag wrote the first of these books in 1978, when she was being treated for breast cancer. She deeply explored the metaphorical meaning then attached to cancer, which at the time was that somehow cancer patients had brought the illness upon themselves as a consequence of pent-up, anxious, fretting, repressed personalities. Psychiatrists attached themselves to this belief system, advocating psychotherapy to disabuse patients of the mindset, the hang-ups said to have led to their cancer. All one needs to do is spend some time at a cancer center (I formerly was on the faculty of MD Anderson Cancer Center) and you realize: this is an illness that strikes anybody, everybody, and there but for the grace of…
We know now this was nonsense, that cancer is a set of errant molecular events with no more moral or mental dimension than a head cold. You would not see a priest for pneumonia, so why see a psychiatrist for cancer? And yet even in our time there are those that still attach metaphors to cancer, often metaphors that stand to advance, to be in service of, other belief systems. I have had deeply religious patients tell me that ministers are trying to draw them deeper into the religious fold, that they’ve been told that unconfessed sin is manifesting itself as their cancer. Those same patients have been propositioned by the execrable practitioners of vitamins, herbs and the alternative, with claims that the illness owes to a lack of naturalness, to vitamin deficiencies, to quaint “imbalances” and that they know solutions (solutions from which these advocates always stand to profit). Mantra recitations do not cure mutations.
As Sontag identifies, other metaphors, still deeply trenchant, have attached themselves to tuberculosis and to HIV. I worry there may be readers here who have cancer, or who will face a diagnosis of it, who view this as the final horrifying intimation of mortality, that a cancer diagnosis is evidence of a body giving up, of a system whose machinery has gone irrevocably haywire, and that things will never be the same. Don’t. In fact, every day you are alive, somewhere in your body, you have cells undergoing transformation to cancer, ones that are conquered by various mechanisms (immune kill, apoptosis). That one of these improbable events serializes and compounds itself….these are, chances have it, things that will befall all of us given world and time enough. Anticipate. Expect. Screen. Pay attention. If and when it appears, play to win and take no prisoners.
In the threads we have discussed personalized medicine, the idea that your genes define you and provide all one needs to know about you. I am a dissident of this view: not all genes are expressed, and even for those that are expressed, we are only now in our earliest stages of understanding what turns expression of those genes on and off. This is the field of epigenetics. What is a four-dimensional printer? It is a three-dimensional printer that in hidden ways confers on the three-dimensional product attributes that allow the printed product to take on structural attributes not strictly reckoned by its three-dimensional printing. In a way, the genome is a 4-D printer: it makes a stew of proteins and RNA, and somehow that stew rises above the DNA sequence and controls it, decides what will be expressed, how and when. We are several Nobels away from a full sense of the science of epigenetics. For eons, people have argued genes versus environment, nature versus nurture. But the truth is it is neither, and both: the products of the genome, in the context of environment (temperature, nutrients, chemicals, and maybe softer constraints, such as social issues), have an amazing way of regulating the genes and how they genes behave. It is neither nature nor nurture. It is both nature and nurture, acting not as independent forces but in combined ways we don’t fully understand. It is what happens at the mysterious intersection of genes and environment that defines us. For many psychiatric illnesses, we now have a sense that it may not be a gene, and may not be a traumatic experience, but may be that the traumatic experience has permanently affected the expression of a gene. Which means that neither psychotherapy nor traditional psychotropes may help it.

In the comedy Trading Places, the fates of an investment banker and a street criminal are swapped to settle a nature versus nurture bet between two rich addle-brained aging brothers. Both lose out, in poetically just ways.
When a lump is found in a breast, when a PSA is sky-high (not 5 but, say, 50), when masses are found at colonoscopy, biopsies are done, tissue is obtained. Why?
That may seem a dumb question. The old answer was: to establish or exclude the presence of cancer. That age may be passing, however. Multi-detector CT imaging, now four-dimensional (the fourth dimension being temperature), already provides so much data corroborative of cancer that biopsy may be superfluous. A former colleague of mine, a radiologist, has become literally a chemist: he is a very bright man, and is actively developing a novel new set of radiologic imaging agents that have such unique ways of binding to tumors (and binding only to those tumors) that they may lead to imaging techniques that totally remainder the need for biopsy. For years I have had recurring arguments with oncologists over one situation: that of the cirrhosis patient with a 5 per cent per year risk of developing hepatocellular carcinoma. The patient’s alfa-fetoprotein is now 1000 (and his testicles are normal), and there is a mass that enhances in the arterial phase of a contrast-enhanced CT scan. If a biopsy of such a mass showed no cancer, would that mean anything? Would you trust that negative biopsy? I say “of course not!” You only do a test when the result will affect patient treatment. And I say, in fact, that we subject that patient to danger by sticking a needle into a safely encapsulated, walled off tumor, spewing its cells everywhere.
There is some necessity of biopsy, of course. As a medical student, I was peripherally involved in the case of a man who had an large opacified mass in his right lung. The mass could not be reached bronchoscopically, but he was bronchoscoped anyway, to look for other evidence of cancer. Washings suggested cancer cells though no cancer was seen. The patient went to the operating room, and his right lung was removed. When that lung was breadloafed up later by a pathologist, the mass proved to be a giant fungus ball, and there was no cancer. The cancer had been in the patient’s left lung, not yet apparent on CT.
A more modern reason for doing a biopsy is the personalized medicine thing: not to make a diagnosis of cancer, but to extract DNA from the cancer, and find what genes, what markers, are present, so as to guide chemotherapy. But in one of the threads, I argued that this has its problems too, and provided references. Tumors are famously heterogeneous, and a needle may have been stuck into a part of the tumor where cells simply lack the markers that will most define the overall cancer’s response to chemotherapy.
But some data, new and very recent, suggest that if one looks at circulating tumor cells, one may get the best reveal. It’s like not looking for Hollywood stars on the internet, but going straight for the Walk of Fame. The “baddest,” the most ill-behaved, most metastatic, most aggressive of the cells in a tumor may be those that pop off into the circulation.
Which is where an interesting new company comes in…
Biocept (BIOC) is a tiny San Diego company ($23 million market cap) that went public on 5 February 2014 with 4.5 million shares. I have taken a position in this company, as risky as it admittedly is, because I regard its circulating tumor cell detection methodology as best of breed.
Biocept has a proprietary method for detecting rare circulating tumor cells using a combination of multiple monoclonal antibodies (with a final avidin-biotin binding step) and complex microfluidics. It does have competition. Janssen Diagnostics (a unit of Johnson and Johnson) has a competing methodology, its so-called CellSearch machine, which several large national reference labs own. An excellent recent study, done admittedly by Biocept, compares Biocept’s method head to head with that of Janssen for detecting breast cancer cells. I have read the full text of this article, and am persuaded that Biocept’s method is superior. It claims an overall detection rate of 89 percent, versus 69 percent for Cellsearch, but for certain specific types of hard-to-find cells, Biocept’s is three times likelier to pick them up. And a success for Biocept in this arena is likely to make the stock catch fire, while broader implementation of CellSearch machines is unlikely to budge JNJ share price. I am long BIOC and have no position in JNJ.
Where Biocept’s method really outshines Janssen’s however, is as regards characterizing the cells it detects. FISH analysis is fluorescent in situ hybridization. Since a strand of RNA or DNA only binds to a complementary strand, it is possible to “probe” a cell isolated by Biocept’s method using diagnostic nucleic acid pieces, look for the presence of oncogenes.
HER2 is an important cancer marker found in about 20 per cent of breast cancer biopsies. The presence of HER2 strongly influences choice of chemotherapy. Women with HER2-positive tumors need therapy with Herceptin, a monoclonal antibody. Biocept published a fascinating study in 2013. Tumors are heterogeneous, and Biocept has evidence that a sizable number of women with HER2-negative tumors by biopsy actually have HER2-positive cells disseminated from the tumor in circulation. This is revolutionary, as those women require and will live longer with Herceptin-based regimens, even though their old-fashioned tumor biopsies said Herceptin would be neither helpful nor necessary. And there is an inherent logic that these cells would be the ones that make in into circulation: HER2 confers aggressiveness and metastatic potential on breast tumors. HER2 is a receptor tyrosine kinase related to the epidermal growth factor receptor.
Biocept is actively developing validated tests for lung cancer, prostate cancer, melanoma, and GI malignancies. Biocept’s methodology is sweetened considerably by the fact that in many aggressive tumors, in the rare cases that circulating tumor cells cannot be found, circulating tumor DNA can be found. Its methodology can detect that, something CellSearch’s cannot do. Biocept’s CEE (for cell enrichment and extraction) can sniff out tumor DNA in a sample, and probe that for tumor markers.
These are the kinds of tumors Biocept’s method can help diagnose and guide therapy for, arranged in order of deadliness:
| Cancer type | Deaths/Year | New Cases/Year | People living with diagnosis |
| Lung | 159,480 | 228,190 | 399,431 |
| Colorectal | 50,830 | 142,820 | 1,154,481 |
| Breast | 39,620 | 232,340 | 2,829,041 |
| Prostate | 29,720 | 238,590 | 2,617,682 |
| Melanoma | 9,480 | 76,690 | 921,780 |
These are statistics about Americans.
Are there risks to being in Biocept? Certainly there are, and this investment is not right for the intensely risk-averse or for a sizable part of your portfolio. My personal view is that its growth prospects are enormous, even extraordinary, even though I rarely invest in diagnostics plays. The technique is new, the methodology the company owns is definitive, and Janssen seems not intent to compete. I feel diagnostic testing with this system will be standard of care within 5 years. But herein lie risks. One is that the FDA has not approved these tests, simply because the field is too new. Approval…for what? For making an initial diagnosis? In lieu of biopsy? As a means of monitoring response to chemotherapy? The method is too new for the company even to know for what it should seek approval from the FDA. The company has many collaborative investigative arrangements with many American institutions, including MD Anderson. Meanwhile, I have spoken with the company and it is quite clear that insurers are reimbursing it for these tests, done at its facility in San Diego, because such testing is preferable to and cheaper than repeat biopsy to guide therapy. So many global institutions now have home-grown studies of ways to pick up circulating tumor cells and looking into their relevance, there is definitely the possibility that some other biotech could yet find a better method. Having read about the methods, however, I am inclined to doubt the would-be competition.
You probably have many questions.
(1) Will Biocept’s method ever totally replace tumor biopsy? That’s too early to call, but it seems probable it will, at least in some cases. The sensitivity of the method is quite high. The company will have to do studies to document the presence of circulating tumor cells in a variety of cancers and get a better sense of how advanced the cancer has to be before it can be detected in blood.
A close friend in Asia recently lost a non-smoking sister, with young children, to lung cancer. Although radiographic evidence screamed that she had cancer (she had a routine chest x-ray, which led to a CT), fear took over, and denial intervened. An unscrupulous traditional medicine practitioner fanned her denial, and promised a cure with Chinese herbs. It could not be cancer, she said. I am healthy and I have never smoked, and besides if it is, I have these herbs. This was a smart woman, a documentary producer, in one of the world’s great cities. There was also the grim reality that to prove it was cancer would involve hospitalization and a grisly procedure, called transthoracic needle aspiration, that would in all likelihood collapse her lung and would in all certainty be agonizing. Except for giving birth, she had never been hospitalized. She was warned by me that she had, at best, 18 months to live if she refused treatment, but that resection would likely cure her. She refused. Her brilliant unconscious was not subject to the rules of transparency and accountability that her brilliant consciousness used, and it slipped off into the dark; it did its calculus and demanded denial as a plan of action. She could not cope with the dissonant truth of cancer. 17 months later, she became quite confused…from brain metastases. One month later she died. Her children are still at home, and motherless. And yet had Biocept’s system been available, a mere blood draw could have provided proof positive of lung cancer, and guided therapy (surgery, drugs, radiation, or what combination). Had Biocept’s method been available, she might now be alive: the data would have convinced her she had cancer that was serious and invasive.
A fascinating just-published study in Nature Medicine, unrelated to Biocept’s efforts, raises the possibility that methods to catch circulating tumor cells are so good now that such cells might be propagated outside the body, in culture or in mice, and then characterized, DNA-assessed, in order to predict response to various interventions. The cells could isolated from a tube of your blood, placed in a mouse, allowed to grow, then harvested and their DNA deep-sequenced.
(2) Will Biocept’s method ever become a screening test for cancer? Many medical professionals reading this may recall a now-nearly-forgotten New England Journal of Medicine paper from when I was in medical school that showed a difference in nuclear magnetic resonance spectoscopy signal between blood of people with and without cancer. The test would give no clue as to type of cancer, of course, and neither could it necessarily be reliable for picking up early stage disease. We will continue to need to screen high risk organs, such as breast, prostate, and colon. Japanese people in Japan will still need screening upper endoscopies at age 40 because of the high risk of gastric cancer in that setting.
Will it ever become a routine blood test to look for cancer? I suspect it will not be priced to make that affordable, and not covered by payers, but if a patient is having other symptoms or labs suggestive of undiagnosed malignancy, it may be considered. Certainly given the breadth and specificity of the testing antibodies Biocept has, it is reasonable to predict that from the blood test, it could narrow down what organ is involved in the cancer.
Again comes the question, however, of whether it will replace biopsy. If the test picked up a cell likely from the pancreas, and the patient has a radiographic pancreas mass, at present oncologists would refuse to treat that patient without tissue from the pancreas lesion showing cancer. With time, with proof of Biocept’s method, with oncology getting farther down the learning curve of this method, however, that may change, especially at academic centers.
(3) If I have cancer, have had a biopsy, and am receiving treatment, does Biocept’s method have anything to offer me? It very much does. Tumor behavior, tumor oncogene expression can change with time, on the basis of what drugs have been given. Those drugs may kill off sensitive tumor parts, and select for resistant cells. Biocept allows determination of this without another biopsy.
The time may come, depending upon price, when circulating tumor cell burden is used as a way to follow response to chemotherapy, much as repeat CT to measure lesion size now is. This fascinating full-text article from 2010 suggests that burden of circulating tumor cells alone is an excellent independent prognostic marker for outcomes in colon, breast and prostate cancer.
Take a good hard look at Biocept, and consider putting some shares in the speculative part of your portfolio. Aegis Capital recently initiated coverage with a buy rating, and a price target of $16, a far cry from the recent $5 share price. Have a go, as I know you will, at Biocept’s recent corporate presentation, where certain particulars are explained in further, if drier, detail. Biocept is certainly the most exciting medical diagnostics company I have ever encountered, and will probably shake up the cancer diagnosis and treatment paradigm like nothing else will. Biocept finds needles in haystacks, but a company this novel, with technology this exceptional, poised for the sort of growth it seems destined for, makes this company a needle-in-a-haystack itself.
This is a discussion topic or guest posting submitted by a Stock Gumshoe reader. The content has not been edited or reviewed by Stock Gumshoe, and any opinions expressed are those of the author alone.
 Of Needles, Haystacks, and the Needle-in-a-Haystack Cancer Diagnostics Nanocap That May Belong in Your Portfolio
Of Needles, Haystacks, and the Needle-in-a-Haystack Cancer Diagnostics Nanocap That May Belong in Your Portfolio


Has this been covered because it is a big deal in the RNA space that broke yesterday.
Arrowhead Research (ARWR) stock was hammered Friday, falling 16% intra-day. The decline may relate to fears about a patent obtained by Alnylam Pharmaceuticals, Inc. (Nasdaq: ALNY).
Yesterday Alnylam Pharmaceuticals announced that the United States Patent and Trademark Office (USPTO) has issued a new patent (U.S. patent no. 8,618,277, or “’277 patent”) in the company’s McSwiggen patent estate.
Quote Dirk Haussecker blog:
“In brief, the McSwiggen patents that Alnylam bought from Merck claims RNAi triggers that satisfy the following conditions:
1) RNAi trigger with strands that have a length that fall within the classical range for siRNAs;
2) At least 10 or more bases (often pyrimidines) of either or both strands have to be one of the following modifications: deoxy, 2’F, or 2’0-methyl.
Depending on the patents there are slight variations as to the strand lengths covered, additional requirements for phosphorothioate linkages etc.
Given the above limitations, it is strange that coincident with the acquisition of the McSwiggen patent estate, Alnylam repeatedly adds in the related press releases that it owns RNAi triggers, including those modified with acyclic nucleotide analogues (–> usiRNAs). By contrast, it would appear that replacing any of those modifications with the very unlocked nucleic acid analogue would be a simple way of circumventing McSwiggen without any destabilizing effect on the RNAi trigger.”
Does this affect BLT?
BLT does not use siRNA … they use ddRNA
Yes, agree with Elliot. BTEBY/BNIKF is up today because of the strife this creates in the siRNA space whilst leaving ddRNAi untouched. Among the biggest reasons I have never owned ARWR is on fear that a dispute like this would play itself out, with one company losing. There are only a finite number of ways to deploy siRNA against HBV, and ARWR and ALNY overlap. The Benitec approach is siRNA liberated by the endogenous actions of DICER. One of the unspoken virtues of the Benitec approach, and one it disappoints me that they have not elaborated on, is that DICER is a sloppy, promiscuous enzyme, and generates dsRNA fragments with siRNA properties from short hairpin RNAs…..but the ds RNAs vary by up to 20 nucleotides in site of 5′ start and 3′ conclusion. Which means that in fact with ddRNAi, you are generating a veritable host of siRNA’s inside the cell, and the big variety of them subverts and overrides virus resistance mechanisms. No one really knew how inconsistent the carving job of DICER is until the Feb 2014 paper by Pfizer/Tacere looked at next-gen sequencing of post-DICER RNA species inside the cell. Moreover, it is quite quite hard for there to be off-target silencing (something RNAi has been accused of risking) when DICER is making such heterogeneous RNAi inside the cell.
David B: you asked above about Conatus. For me personally, this stock is a canned laugh track for biotech investing. We have discussed it some on one of the threads. its sole agent is emricasan, a pan-inhibitor of caspases, intracellular proteolytic enzymes. Their aim is to use this to hobble apoptosis….as if apoptosis is the enemy. It is not. Cells constantly scan themselves and any hint of an immortality-conferring error activates the caspase suicide pathway. They are giving this drug foolhardily to people with serious liver disease, many of whom are at high risk of hepatocellular carcinoma, and in my view are predisposing them to eventual tumors because the drug rescues cells that should be let to die.
I feel that one can tell much about a company by the quality of clinical trial decisions it has made before. To wit, this company squandered hundred of millions of investor capital chasing a matrix metalloproteinase inhibitor as a therapy for HCV. There is no reason on God’s green earth such an agent, which has no action on viruses, which modulates the matrix in which cells live, should have ANY effect on HCV. Anyone with an iotum of intelligence could have told them that before they started this fool’s errand trial. But they did the trial anyway….again and again! And saw——brace yourself, hold onto something!—NO EFFECT on HCV.
Don’t reward these dunces by investing in Conatus. I will be happy to send an autographed dunce cap to the CEO. He richly deserves it. Remember that film “The Name of the Rose” based on the Umberto Eco novel? There is this weird polyglot character drawn from somewhere beyond Quasimodo, and he always speaks in cognate garbage. it is what comes to mind here…..estupido, estupidamente, loco, dumkopf. This is an outfit cut from a slightly less tattered piece of the same bolt of cloth as PVCT….a bunch of guys lost in groupthink, stuck in mass delusion….folie a deux times 10. This drug is a bozo decoction and the company will crumble.
Sounds like a very good one to short based on its recent upward movement. I thought this one was likely a stinker since you hadn’t touted it and it’s in your wheelhouse.
OK. Someone with good spreadsheet skills might want to mark a definite no by Conatus.
This will probably not be the last time the company crosses into our line of sight.
Here’s one that seems to have all the makings of a flim-flam squall, a company I have bird-dogged for a bit. Recro Pharmaceuticals (REPH….not to be confused with Retrophin). Right off the bat, the name strikes me as screwball…”Recroom”? I wondered if this is some virtual company being operated via file folders atop someone’s billiards table. And that may not be far off the mark.
This company claims it is commercializing intranasal dexmetomidine for pain. This drug is a central alpha2-adrenergic receptor agonist that has been used iv for sedation in ICU’s and for interventions. Sedation, however, is not the same thing as analgesia, and I see no evidence this has analgesic properties. The drug is not controlled, which preliminarily is unsurprising because based on how it works, it should not be habit forming. That is one corner of whether a drug is controlled. I suspect it will become controlled however, as it alters the sensorium so much that it easily could be exploited for getting high. Meanwhile, an anxiolytic for pain makes no sense. If you sprain your ankle, you do not pop a Valium for it.
This company claims to be have done and be planning multiple trials. Multiple queries of multiple databases says nyet to that, however. I cannot find a single registered trial. Or publication, or presentation.
The company’s physical address is a shopping mall in Malvern, PA, and is identical to that of Malvern Consultancy Group, which claims to provide advising and clinical development services to pharma companies. All well and good, except that among MCG principals, there is not a single doctoral level person….no PhD’s or MDs. The group president is an elderly lady who began her career as a drug representative for a big company. Recro’s principals are all MCG principals.
The company lists Argot Partners, a Park Avenue firm, for IR. A query I made to Argot about the trials question has not been responded to.
This company’s recent IPO was lead-underwritten by Aegis, which has a strong buy rating. That’s the company’s only analyst rating. Aegis underwrote and has a strong buy on Biocept. I do not think Aegis is P&D, as I feel Biocept is a fine company. But I now have serious questions about the worth and legitimacy of an Aegis endorsement.
I am deeply concerned REPH is just a fleecing operation: a gambit by MCG to bring in a few million bucks for itself because I seriously cannot envisage a serious pharma firm hiring this rinky-dink group of Keystone Cops to help with drug development. I predict there will be “unexpected obstacles” and, oh, gosh darn, “regulatory delays” to getting the dex trials going, and that trials will never happen. I think that the IPO proceeds are to keep MCG in top shelf organic coffee, doughnuts, nice lunches, and posh liquor cabinets for the MCG staff and offices. I honestly call upon the SEC to protect consumers and check this company out….I think MCG is there to create the illusion of paperwork and regulations and goings on but that there is absolutely no intent to develop a drug here. Anyone in these shares might want to quietly vacate.
Dr. KSS: Thanks for the info on REPH. I’ll keep an eye on this one. All we need to do now is wait for one of these MCG guys to show up from no where on this forum.
RE REPH: Broadfin Capital owns 8.2% and Sabby Management 3%. Interesting.
Paul Edick, the CEO of Durata, bought 3000 shares yesterday and holds about 45,000 shares.
What it at market or some discount deal?
Market from what I’ve gathered, he’s bought at mkt prices before.
Some large block owner of DRTX dropped 751,000 shares, worth $11.5 million, on 12 June. I have looked through SEC filings but still cannot discern who it was. If we knew who, we could discern why, perhaps.
Dr. KSS, sorry I cannot pinpoint either. Is the 751k block activity visible??
Stockwatch.com DR. KSS seems to look at block trades and may show who if you navigate to Analytics->Block Trades and then there is a blue help link that explains the search info it displays. But I don’t have membership, It looks like you can grab a 30 day trial.
How bout that intra-day turnaround for Durata?!?!?! Wheeeeeeeeee!!!!
David B: Ocera Therapeutics (OCRX) is one that has come up before here, for its iv drug ornithine phenylacetate for cirrhotic portosystemic encephalopathy. This company has been abused, and it is down from highs of almost $20 to about $7.50 now. This drug will never shake the sheets and bust blocks, but it will get approved and find use for the occasional sick cirrhotic inpatient who is confused and not tolerating oral lactulose (nowadats I usually give such patients serial lactulose enemas, but that is quite messy). I can easily see this stock moving back to, say, $12-13 from here over the near term. Hyperion has a competing agent, but I am not sure they are pursuing a formal FDA label indication. Ocera’s drug won’t work of course in renally insufficient cirrhotics, which are common. It will always be a niche item, but it will get to market I feel.
Lou L: the only way REPH could have escaped the trial databases for demedetomidine is if they have not submitted an IND. Possible but not likely. If they have not submitted an IND, the trials would have to be conducted only in Pennsylvania, as part of the reason for IND is that it incurs legality for interstate shipment of investigational drugs. If they are doing this without an IND, that is a real waste of investor money, I feel, as the FDA will snub the work. Meanwhile I did find trial records of at least 15 universities in 15 countries doing clinical trials of intranasal dexmedetomedine compounded on premises by pharmacies. Keep this in perspective: I can find proof a trial is going on in, say, Turkey, but not evidence of a trial in America by an American company wanting you to buy its traded-in-America shares. I will never hear back from their IR firm, but if I do, what I will be told is what Indiana Jones was told at the end of “Raiders”….”the ark is being studied by TOP men…” The trial is going on at TOP places, I am sure. They can’t be named, but they are TOP.
Dr. KSS, I am tempted to short some shares, but I don’t want to bet against the big boys like Sabby.
TOP= The Official Procrastinators. Bureaucracy is heavy laden with them. fa
Genuinely superb piece, wise and profound! I wonder, however, what the right response to the poor guy whose agreeable gin-and-tonic (with tasty lime!) was interrupted by the message that his PSA had risen from about 4.0 to maybe 4.5? The Biocept assay, as you pointed out, is not yet being put to front-line diagnostic uses. My guess is that the physician would say, “Let’s do another PSA in six months or so.” But supposing that it had doubled, say from about 2.0 to a bit over 4.0 in the same interval? Would a biopsy at that point be advisable? A biopsy would spot tumors within the prostate capsule, and provide a pretty good picture of the spread of the cancer in the capsule and the location of the tumors (but not whether the cancer had spread beyond the prostate itself). Would the Biocept assay be able to furnish comparable information? For that matter, in the case of prostate cancers that have not yet escaped the capsule, is it known whether cancer cells are present in the blood? By the way, I can say from personal experience (having been through this scenario myself 12 years ago, including radical prostatectomy, with great success) that a prostate biopsy is mostly no big deal. Don’t be scared, guys!
Another issue potentially affecting the fortunes of BIOC is the current anti-testing trend – no PSA testing for men, no mammograms for women in their 40s, no further cholesterol testing for people once they have been put on statin therapy on the basis of a simple algorithm. This is mass medicine as practiced by robots.
The projected Biocept assay promises the direct opposite, an individualized procedure focused on minimally invasive patient care. I am solidly in that camp.
Hi Michael, Wow 12 years! That’s great. What are your recent PSA readings?
Hi Michael, Glad you are 12 years out. I also went through this 14 years ago (seed implants and beam radiation). For biopsies you can have the area numbed which I strongly encourage. I didn’t and it was not a good experience. You hit on a good point. Current wisdom is that the PSA doubling time is more important than actual PSA numbers which is some cases makes it important to have a good base line.
If your doctor suspects your cancer has spread, there are various tests that can be used including: bone scans, CT scans, MRIs, ProstaScint scan, and lymph node biopsies.
Respectfully, I would suggest that it is important to distinguish between the universal use of screening tests for otherwise apparently healthy individuals, and diagnostic tests. They are not the same, and it makes sense to me that the criteria for recommending the former would differ. I am persuaded by the evidence that suggests it is not good policy to recommend that all women between 40 and 50 receive screening mammograms, and it seems to me that the jury is still out on the advisability of universal PSA screening. Neither procedure is without its risks and false positives, which any individual without particular risk factors that would justify the screening tests should take into consideration. I don’t believe the issue is as simple as you make it out to be.
KPTI (Karyopharm Therapeutics) up 65% today and still pushing. Supposedly on good phase 1 data.
http://www.streetinsider.com/Corporate+News/Karyopharm+Therapeutics+(KPTI)+Gains+Following+Phase+1+KPT-330+Data/9581646.html
I am still catching up on posts here and the other threads after 2-3 days of viremic mattress-soaking febrile flop sweats. I think the issue of FDA approval for BIOC’s tests has come up. Let me be clear: they are not FDA-approved. They are covered by payers but not FDA-approved. My contention is that the testing method is new enough that it is quasi-investigational still, but also being hailed as a way of avoiding biopsies (which are quite expensive). I view it as I do a pathology testing modality: if someone identifies that a certain vital dye is good for staining a carbohydrate antigen on tumor cells, a pathologist does not need FDA approval to use the stain. If BIOC were saying, we can tell whether or not you have cancer, well that probably would need FDA approval. They are saying that our test can follow and prognosticate cancer, and they are amassing data to support it. I suppose there is always the freakish possibility that Margaret Hamburg could get a wire up her backside and order BIOC to halt what it is doing, and frankly in light of the weirdness that befell Orexigen over Contrave (note to all companies; do NOT seek approval for combo drugs…..the FDA is just against combos as it was with QRx) I cannot exclude some tiny chance of that happening. But I don’t think it will. This new technology will have to write rules for how it is to be used, and not let the system do that. I feel this issue, of acceptance, is taking care of itself by intellectual acclaim and fine studies. The FDA would be brazenly idiotic to stand in its way and say, No blood testing….we want painful, bloodletting biopsies for all!
Someone above asked if this is like another company with a “blood test for cancer.” No, it isn’t, in no way even remotely similar. That company is flimflammy, testing for one of many nonspecific markers for cancer, and can tell you nothing else except that you might have cancer….and will provoke many pointless million dollar snark hunts. Cancer does not just up and happen in some occult way in which it tries desperately to hide. It makes itself known with features and symptoms that a doctor can pick up. A blood screening test for a protein released by cancer cells is just about utterly worthless for clinical medicine because there is no management it will affect, no difference it will make in patient care. With Biocept’s method, it can tell you what tissue the cell is from, and all about its molecular characteristics and what drugs it will respond to.
Janssen calls its method CellSearch, but to me Biocept’s method is CellFind. The former snoops and missing many cells. Biocept always nabs the wafting-by tumor cell, however, as it is hard for one to escape its antibody dragnet and flow funnel chute.
Excellent clarifications. I’m not in yet but this looks like a good long term buy and hold. It likely wont set the world on fire over the next 12 months but it will do very well over the long haul.
Clomu: Karyopharm is pretty interesting. I am surprised that none of the systems I have in place to sniff out insider buying picked up on this one, but it seems they didn’t. Today’s price movement is not warranted, as this is a tiny study, and one that cannot rightfully even say anything about efficacy. They will have to do a 1b to find MTD. This gap will fill. I will look deeper into it, however, as it may be worth getting in on when it comes back to earth. 1,5 million shares bought yesterday by entities prior to the trial data announcement. It is now 98 per cent spoken for, but with about 15 per cent short interest, which will likely rise from here. Its nuclear pore compounds have a long long way to go.
Michael: the most elemental, fundamental question Biocept will have to answer or help answer is sensitivity of its approach for a given tumor size (what percentage of people with the cancer have CTC’s by our assay). Metastasis risk is proportionate to how many cells are in a primary, and tumor size and number of cells are proportionate to each other. In prostate medicine, there is a general guideline that an increase of PSA by more than 0,2/12 months is a cause for concern. PSA is linearly proportionate to number of cells in the gland, and so for the PSA to go from 4 to 4.5 implies more than 10 per cent more cells than last year, all other conditions at time of determination being equal. The first derivative hints at a mitotic process.
WHat the company seems to be doing is partnering tightly with cancer centers to be running these assays en masse on cancer patients, and just doing that as a mass means of getting data: for a given tumor size and kind of tumor, what are the chances of finding one cell in 10 cc of blood?? Biocept, several universities are several other companies have formed a big giant investigative prostate cancer consortium to re-define the diagnosis and management of the disease….new markers, new thresholds for what those markers mean, and I feel that from this one being prostate ca effort, a clear sense will emerge about at what levels of what marker a test for CTC’s becomes relevant. I do not think the marker of the future will be PSA, At one time I thought it would be PSMA. It could prove to be GRP78.
Anyway, I think clear vivid numerical answers to the fine questions you ask, about sensitivity and specificity for a cancer at a given size and stage of the cancer…I believe that clear answers will be on hand in 5 years. Above all else, though is this: I feel a CTC assay is BETTER than biopsy because to escape and metastasize, a tumor cell must have powers of aggression beyond the average tumor cell. It is already clear that more useful data comes from CTC than from biopsy. By CTC, you are catching the wiliest, most bad-acting members of the cancer, while many of the cells in the needle biopsy are merely indolent. It makes me think of a junkyard with a pack of wild dogs. Only the meanest and most rabid ones make it over the fence to hound you. Only the meanest, deadliest tumor cells make it out of the capsule many tumors live in and into circulation. Only good tasting tuna gets to be StarKist, and only truly deadly cancer cells (the ones you want to target with chemo) make it into circulation. Chemo needs to be dialed in, selected, based on what is in blood, not what is in the biopsy. That is what the data now are saying rather screamingly. Insurers realize that if you give the right ‘mo the first time, fewer ‘mo courses will be needed, so it is cheaper for them to treat based on CTC assays.
The CTC issue, of course, is why so many tumors wind up in liver. It is the great filtrative reticuloendothelial organ. Most blood flow ends in capillaries that soon size up again to venules. Not so in the liver: the portal venous and hepatic arterial inflows merge to form hepatic sinusoids, through which blood percolates along sinusoidal-lining-cell lined spaces and where a CTC easily deposits, gets caught.
Right now, every assay BIOC runs is in some way contributing to a living textbook on CTC’s. They want to know size of tumor, what drugs have been given, duration, stage….they are databasing everything they do to construct a strong case for the importance of this method. I really think some of the most relevant papers ever to come out of medicine will be generated by BIOC in the next 5 years. I think it will revolutionize cancer management enough that the Swedish Academy will have to consider honoring it, but it may have to give posthumous prizes to Ashworth and Trousseau because no single group of 1-3 people has advanced the field as they did.
Lou: I may try to call company HQ or the IR office in NY. At this point I kind of want to see what their pain threshold is. When people are scamming, I enjoy twisting the knife. The IR people have had ample time to get back with me, and I was quite friendly to them in my letter. I made no accusations and stirred no ill will. To be honest, my sense of MCG is that if you have to interact with them, you cringe….you go, Oh no, you guys are going to TALK me to death. So, underwriters are happy to sign off on it just to shut them up. I could be wrong. I think these are people that can bury you alive in hooey and paperwork and not produce the goods. They are only too happy to give you three days of presentations about Recro and never make a point, never tell you what you came to hear. I imagine them sounding like that blahBLAhblah voice of Charlie Brown’s teacher.
Hi Dr KSS-glad you’re feeling better. thans for another thought provoking article. I’ll post my commnet here, since this seems to be the new thread. Thanks for your response. In terms of glutamate, pateints takng Neudexta don’t seem to get depressed. It is so plentiful and ubiquitious in the brain that some will always be there. This drug lowers the levels but does not deplete it. I have limited experience, but information f rom a colleague with lots of experience and what I read suggests that depression doesn’t seem to be a problem. In PBA, the patient is neither depressed or elated when they emote. But after taking Nuedexta they report no change in mood but decreased unexplained and spontaneous affect. As for addiction, from what I understand it is the metabolite, which is blocked from forming by quinidine, that causes the high, so addiction is not a problem. The low dose of quinindine should not cause problems for anyone foolish enought to try to take too much. But if it can treat the negative symptoms of schizophrenia and curb the aggression in Alzheimer’s that would make this drug a big sucess.
Here’s a link for the Senesco ASCO poster:
http://meetinglibrary.asco.org/content/98553?media=vm
Jer Vic: that’s quite good news about SNTI really. Hope Thomas Tucker is still reading. No bad data, they are still just dose finding and have not hit a maximum tolerated dose yet. Which means the drug is safe—-it was predicted to be quite safe, but good to know that it is. This should not have beaten down share price for this company at all. Cancer phase I’s are always dreary because you have to dose, watch, and then dose escalate. Most phase I’s are about finding whether a reasonable dose is toxic. In cancer phase I’s you start low and dial up to a point where either you are beyond where you will dose clinically or the patient begins to experience something adverse (vomiting, falling counts, a bump in liver tests or creatinine). They are still hunting for MTD and this could take a while yet. But this is good for Senesco!
This is becoming the summer of Chikungunya:
http://www.reuters.com/article/2014/06/12/us-usa-health-mosquito-idUSKBN0EN2F820140612
Dr. KSS, Thank you for such wonderfully written and informative articles. One of the fun things about investing is how much you learn. I am wondering if you have any thoughts about INO? I see someone had suggested shorting it. Ouch. I have some shares in it. Is it worth a long hold? Do you think their approach can work?
Thankyou again for your articles. You are one of the clearest medical teachers I’ve read!
Peter and others: Minerva Neurosciences is a company planning an IPO, not yet priced (it will go public as NERV). They have a sigma-2 receptor-acting agent in phase II for schizophrenia, which interests me because the sigma receptors seem uncharted waters. They also have a combined dopamine/serotonin reuptake agent (as I recall) for depression, and an orexin-2 antagonist for insomnia. The last drug would compete with Merck’s suvorexant, which last time I checked (it’s been a while) was bogged down in DEA deliberations about whether it should be a controlled substance or not, though it seems to have efficacy, as I recall. I don’t have a sense at all of when this one will price. I suspect it is at least a month away. While they will not precisely revolutionize psychopharmacology, I do see them as not a company to dismiss, maybe like a budding Sunovion.
thanks for the info as it has dual interests for me-professional and financial. Please keep us posted and in the meantime I will do some research.
Things have been quiet about Minerva (NERV) on these boards since June as far as these boards have been concerned. Since then, Minerva has issued stock. In addition, insiders have been buying more of their own stock (no sales). Also see that Johnson and Johnson is a 10% owner. Schizophrenia drug finished with phase 2a, starting phase 2b in 1st quarter of 2015. In addition to the schizophrenia , depression , and insomnia studies you mentioned, I see from their website that they also apparently are testing a drug for Parkinson’s disease. Any further comments from Dr KSS on this earlier find?
Stephen,
Re: NERV
Thank you for the excellent post re Minerva. I suggest that you repost it on the current KSS thread at
http://www.stockgumshoe.com/2014/11/microblog-payola-from-portola/#comment-3227259
Otherswise, it may not be read by Dr. KSS.
Thanks.
Nice herding Lawrence. Approve.
Dave: it’s odd, because on the charts I do not see such a sale either. On e-trade there is a tab for insider activity, and under that tab, another tab for ownership. Under the latter, they list large block owners and changes in b lock ownership, and show about 751,000 shares liquidated among block owners yesterday. They do not say who. They do not say for sure that it was a single owner either. I wondered if it could be some sort of nonfloat transaction, but DRTX has a difference between float and outstanding shares of only about 200,000. The way the data is presented, it is unequivocal that what is meant is a large block owner transaction.
Dr KSS: Added another reply to your DRTX 751k about Stockwatch.com.
CAVECALLING: ANY GS MEMBER BY CHANCE HAVE A MEMBERSHIP to Stockwatch.com
or ANOTHER AVAIL TOOL TO LOOKUP THE 751K SHARE TRADE (BY WHOM) OF DRTX OF JUNE 12??
THANKS IF YOU HAVE CAPABILITY OR CAN PROVIDE LEADS….Dave