[ed note: Michael Jorrin is longtime medical writer who has been sharing his thoughts with our readers as “Doc Gumshoe” for several years (he’s not a doctor, I gave him the name). He generally covers medical and health news and sometimes health promotions and hype, but he rarely opines about investments or specific stocks. All of his past commentaries can be seen here]
Sometimes is seems as though the answer to most questions is “that depends.” There are at least two ways that we can look at that question. On the face of it, our concern should be based on the specific threat. If the side effect is a mild headache or a short spell of stomach upset, but the drug knocks out a severe infection, no big deal. But if the drug is meant to treat a relatively trivial disorder, and the side effect could be fatal, then it’s a very big deal indeed.
However, there’s another whole dimension to the consideration of side effects, and it is this: many people are dissuaded from taking a drug that could be highly beneficial in treating or preventing genuinely serious diseases or conditions, based on mostly exaggerated fears of side effects. Fear of side effects, in turn, can be exaggerated in several ways. Some side effects are exceedingly rare, but decidedly severe, and people don’t want to take that chance, even if it means forgoing the highly likely benefit they would experience from taking the drug. Some drugs are associated with a long list of possible side effects, most of which are relatively innocuous, but the list itself intimidates persons who would likely benefit from taking the drug.
What does the list of side effects really mean?
Here, to vex and confuse you, is the list of side effects in the official prescribing information (PI, also called “package insert”) for Lipitor (atorvastatin), which, as you probably know, has been one of the most-frequently prescribed drugs on Planet Earth. I’m using Lipitor as an example of how to interpret the side effects listed in the PI, which are also the ones that are mentioned as possible side effects in the direct-to-consumer (DTC) advertisements and the material provided to patients along with the drug.
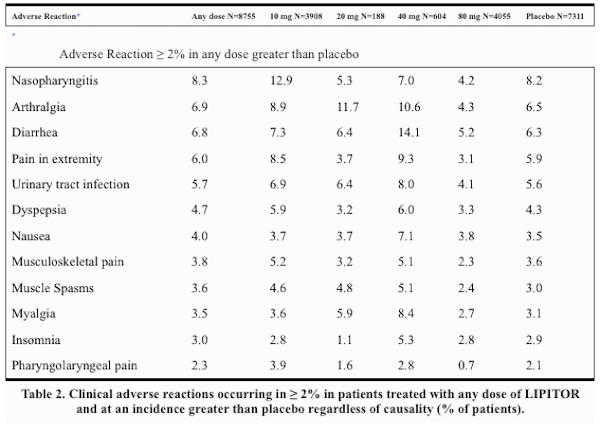
The side effects, also called “adverse reactions,” are listed in order of frequency at any dose, based on the results of the clinical trials submitted to the FDA for approval of the drug. The side effects on the list were those that were observed in at least 2% of the subjects in the study and also were observed at a higher rate in patients taking the active drug than in placebo-treated patients. Thus nasopharyngitis takes the lead, this being the medical name for the condition that most of us would refer to as “coming down with a cold,” i.e., inflammation of the passages in the nose and the back of the throat.
As you see, 8.3% of 8755 subjects in clinical trials who took any dose of Lipitor experienced this symptom. Oddly, the highest percentage (12.9%) was observed in the 3908 patients who took the lowest dose, 10 mg. But not so oddly, 8.2% of patients taking placebo instead of Lipitor also had nasopharyingitis, so one can’t really say that Lipitor was the cause of this not very severe side effect. But at least in the group of subjects on the 10 mg dose, the 12.9% incidence of this symptom was more than in the placebo group, so nasopharyngitis qualifies as exceeding the placebo incidence and leads the list of side effects in the PI. Another odd bit of data is that the lowest incidence of many of the listed side effects was seen in the patients on the highest dose, contradicting the usual truism that “the poison is in the dose.” Could it be that there is an unrelated (pleiotropic) benefit with Lipitor? There’s a fair amount of data on that topic, and I’ll check into it in another post.
But in assessing the risk that an individual will actually be affected by side effects, a number of factors need to be considered, not the least of which is that our own physiology will have a large effect on whether a particular side effect will cause us a problem. Some persons are more susceptible to disruption of a particular body function. For example, persons with “delicate” digestive systems may be more likely to experience diarrhea or dyspepsia or nausea than individuals possessed of “iron stomachs.” But the latter group of individuals may be more affected by other adverse reactions to which they are more susceptible.
And, of course, there’s always the question of the reverse placebo effect, which some observers have labeled “nocebo” effect, meaning that the mere fact that they have taken a pill of some kind, whether an active drug or a completely inert “sugar pill,” somehow persuades them that the stuffy nose or joint pain or diarrhea they are experiencing is due to the pill. But of course, we know that we experience many of those adverse reactions at some point whether or not we have taken any pill at all.
The prescribing information also provides a list of other side effects that have been observed in clinical trials, but which did not meet the standard of occurring in at least 2% of all treated patients.
Body as a whole: malaise, pyrexia; Digestive system: abdominal discomfort, eructation, flatulence, hepatitis, cholestasis; Musculoskeletal system: musculoskeletal pain, muscle fatigue, neck pain, joint swelling; Metabolic and nutritional system: transaminases increase, liver function test abnormal, blood alkaline phosphatase increase, creatine phosphokinase increase, hyperglycemia; Nervous system: nightmare; Respiratory system: epistaxis; Skin and appendages: urticaria; Special senses: vision blurred, tinnitus; Urogenital system: white blood cells urine positive.
And then there’s a list of possible drug interactions. Patients taking cyclosporine as well as certain drugs used in treating HIV and hepatitis C are advised to avoid Lipitor completely; patients taking certain other drugs including the antibiotic clarithromycin and antifungals including itraconazole are advised to use Lipitor with caution and at the lowest dose needed.
By far, the side effect of greatest concern with Lipitor, as well as with other statins, is in the general category of muscle involvement, and the single most serious side effect is rhabdomyolysis, which essentially is a destruction of the muscle fiber itself. Before we go too far down that scary path, let me point out that statin-associated rhabdomyolysis is exceedingly rare – about one case in 20,000 patients – and that the great majority of cases of rhabdomyolysis are due to other causes, such as severe physical injury. This condition is not particularly difficult to detect; among other things, it is preceded by severe muscle pain, and fairly early in the course of rhabdomyolysis, traces of the depleted muscle protein discolor the urine. At that point, the patients simply stop taking the statin, and the condition resolves.
However, rhabdomyolysis, along with those lists of possible side effects, can certainly scare people away from taking Lipitor and other statins, regardless of the potential benefit that they may derive from these drugs.
But there’s more. In considering the impact of side effects, an important category is possible drug interactions, either with other drugs, or in some cases with food and drink. The PI points out that Lipitor is metabolized in the liver, by means of the hepatic cytochrome 3A4 in the P450 system, dubbed CYP3A4. What does this mean? It means, for a start, that Lipitor is not excreted unchanged in the urine, as many drugs are. Instead, it is transported to the liver where it is dismembered, inactivated, and shuttled along to the bile duct for excretion in the feces. CYP3A4 is an enzyme that does that essential work. But that enzyme does a lot of other work as well, metabolizing other drugs and stuff in the bloodstream that needs to be gotten rid of. And there are ways that the activity of this enzyme can be affected. If other substances are employing that particular liver enzyme as a means of egress from the system, any particular drug – e.g., Lipitor – may just have to wait in line. Meantime, the concentration of Lipitor, or of any other drug that goes through that exit, continues to increase, and along with that increase, the likelihood that the patient taking the drug will experience an adverse effect.
One of the things that affect the CYP3A4 enzyme is grapefruit juice. Thus, it is theoretically possible that taking Lipitor along with enough grapefruit juice will increase the concentration of Lipitor in the patient sufficiently to trigger an episode of any significant side effect, including that really scary one, rhabdomyolysis. Possible, I say, but extremely unlikely.
However, side effects related to P450 system function are not uncommon and have in some cases led to the withdrawal of some drugs from the market. Here’s one:
Hay-fever pills that could kill you!
A sensationalistic headline, more suited to a sleazy supermarket tabloid than to Doc Gumshoe’s careful, respectable postings. For a start, let’s tone down the headline – it should be “Hay fever pills that could have killed you,” because they got yanked off the market when the dangerous data came out. The particular antihistamines were Seldane (terfenadine), and Hismanal (astemizole). Both Seldane and Hismanal can have an effect on heart function. At the normal dose for the treatment of allergic reactions such as sneezing and itching, the effects on heart function are insignificant. But overdoses of these antihistamines can affect a phase of the cardiac cycle identified on electrocardiograms as the QT interval – the phase when the cardiac muscle cells take on an electric potential by the rapid intake of positively charged sodium ions, then discharging this potential resulting in the contraction of the ventricles, and then repolarizing through the outflow of potassium ions. Seldane and Hismanal, at high doses, can block or delay this last action, an effect called delayed after-repolarization. The result can be an extremely dangerous and sometimes fatal cardiac arrhythmia called torsade de pointes (“twisting of points”), which describes the bizarre ECG tracing on which none of the heart’s phases are where they’re supposed to be.
Many cases of torsade de pointes (TdP) are congenital, and some are known to be caused by a class of antiarrhythmic drugs that are intended to treat tachycardia. But the incidence of TdP related to antihistamines came as a considerable surprise and shock. As I mentioned above, at normal doses the problem does not arise. To be sure, overdoses of just about any drug you can think of will result in bad reactions, and sometimes very bad reactions indeed.
However, the TdP events in patients taking Seldane and Hismanal were not due to overdoses. They were due to the effect mentioned above – inhibition or outright blockade of the channel through which the antihistamines exit the body. Both Seldane and Hismanal are metabolized by means of that P450 system in the liver, and specifically by the CYP3A4 cytochrome. But when that means of egress is unavailable, the concentrations of those drugs goes up and up, so that the effect is exactly the same as if the patient had taken a colossal overdose.
"reveal" emails? If not,
just click here...
What makes that exit route unavailable is the inhibitory effect of another drug. Drugs that inhibit CYP3A4 include common antibiotics of the macrolide class, such as erythromycin, and common antifungals, such as ketoconazole. This is a particularly sneaky and dangerous kind of side effect. People take macrolide antibiotics and antifungals for a large variety of common disorders. And it is not in the least unusual for a person who is taking erythromycin, for example, to develop a mild itch in response. And in response to the itching, what’s wrong with taking an antihistamine? Seldane and Hismanal were big steps forward in the antihistamine field, because they were non-sedating, unlike Benadryl. But the combination of those CYP3A4 inhibitors and those antihistamines that needed those same liver enzymes to exit the body could and did result in a certain number of cases of severe and near-fatal torsade de pointes, and to the banishment of Seldane and Hismanal.
The number of drugs and other substances that we eat or drink which have an effect on the P450 system is very large indeed. And the kinds of effects also vary quite a bit. In some cases, a person experiences difficulties getting a sufficiently high concentration of a drug to achieve the desired effect. A practical means of raising the drug concentration is to administer the drug along with something that slows its elimination. For example, some patients are advised to take cyclosporine with grapefruit juice, which inhibits the enzyme that metabolizes cyclosporine, thus increasing the cyclosporine concentration to the desired therapeutic level.
And then there’s a possible reverse effect.
A drug interaction that could prevent oral contraceptives from doing their job
The chemical entities that interact with the P450 system are grouped in three buckets. One general bucket is what we call “substrates” – substances that are metabolized by one of the hepatic enzymes and use that pathway to exit the body. The more substrates of a particular cytochrome are trying to squeeze through that passageway, the longer it takes for any particular one to get out. But then there are some substances that actually inhibit a pathway. The particular pathway we’ve been talking about, CYP3A4, is inhibited by quite a number of drugs, including some commonly-used cardiac medications such as diltiazem and verapamil, omeprazole (Prilosec), and many others.
But some drugs do the opposite. They can induce, or accelerate, the activity of some hepatic pathways. For example, that same pathway can be accelerated by anti-seizure drugs such as Tegretol (carbamazepine) and Dilantin (phenytoin), by Phenobarbital, by rifampin, an antibiotic used to treat tuberculosis, by ritonavir, used in HIV therapy, and, not least, by alcohol. So instead of increasing the concentration of other drugs that are metabolized by that pathway, they decrease the concentration, and, therefore, the efficacy.
As it happens, oral contraceptives (OCs) are metabolized by that same hepatic pathway. If a woman takes a birth control pill and at the same time is taking a high enough dose of a drug that dials up the activity of that pathway, the little pill might not work. There have been unplanned pregnancies that may have been caused by such interactions. It’s difficult to establish a precise chain of causality, but the OC makers are keenly aware of the potential for failure due to such causes.
Are the warnings about alcohol just markers of excessive caution?
Not always, by any means. As described above, it’s certainly possible that in some cases alcohol speeds the metabolism of some drugs, weakening their effectiveness. In such cases, it makes sense to lay off the booze; you’re taking the medicine, you may as well get the benefit. But in some cases, the combination of a drug and alcohol results in truly nasty reactions. In previous Doc Gumshoe rants, we’ve heard about the disastrous consequences of chronic acetaminophen (Tylenol) along with robust levels of strong drink. The results can be liver failure, requiring in some cases liver transplantation. That’s just one instance of a drug-drug interaction involving alcohol, so to speak.
Another nasty one is the so-called “Antabuse reaction.” Some persons who would like to stop drinking alcoholic beverages take the drug disulfiram, marketed as Antabuse. When a person has disulfiram in his/her system, even a small amount of alcohol produces a severe reaction – nausea, violent vomiting, severe headache. Individuals who have experienced the Antabuse reaction become extremely wary of indulging in the tiniest drink. But it’s not just disulfiram that triggers this type of reaction. Antibiotics such as cefotan and metronidazole (Flagyl) can produce the same effect, along with a drug of the sulfonylurea class, chlorpropanide, which has been used to treat type II diabetes.
It’s not always easy for a patient to tell from reading the package insert whether the “limit alcohol” warning should be interpreted as “even a small amount of alcohol can trigger a dangerous reaction” or “this drug works better if you stay away from alcohol.” Presumably, the prescribing physician has a responsibility to warn patients about potentially serious interactions. But physicians are busy and may forget. It’s a good idea to have a look at the “Warnings” and “Contraindications” sections of the PI if you have any concerns. Those need to be taken more seriously than lists of possible side effects.
How about drugs that affect the immune system?
Those would include steroids and other immunosuppressants. These drugs dial down the activity of the immune system in various ways. For example, a class of drugs that are highly effective in dealing with such diseases as rheumatoid arthritis and psoriatic arthritis are the tumor necrosis factor alpha (TNFα) inhibitors. I mentioned these agents in the previous Doc Gumshoe as showing promise (perhaps!) in treating Alzheimer’s disease. But a potentially serious side effect with these drugs is that if the patient has latent tuberculosis or latent herpes zoster, which is being kept in check by the immune system, treatment with a TNFα inhibitor may reactivate the disease.
Before embarking on treatment with a TNFα inhibitor, patients should be tested for latent TB by means of the Quantiferon test, which can detect not only current TB infection, but the presence of TB pathogens that have been encapsulated in the body long after the end of active symptoms. Some people have been infected with TB, never developed symptoms severe enough to require treatment, and may still have the pathogen in their bodies. Therefore it is not always enough for a physician to go by the patient’s history in determining whether to test for latent TB. Persons who have lived their whole lives in regions where TB is uncommon are a safer bet, but TNFα inhibitor treatment is a lifetime commitment in most cases, and reactivation of latent TB is a risk that should be avoided.
An intuitive fear comes with TNFα inhibitors: if we’re inhibiting the action of an agent in the body that attacks tumors, are we not increasing the risk of cancer? Happily, the data says no. The only form of cancer that increases with these drugs is non-melanoma skin cancer, and the increase is very slight.
Steroids such as prednisone present a different and more complex problem, which I cannot do more than mention in this piece. The short answer is that they increase the risk of any infection, and if a person has even a mild fungal infection, e.g., onychomycosis (toe-nail fungus), the fungus is apt to flourish. In general, steroids are immensely valuable. They are beneficial in treating a great many ailments and virtually indispensable for some, but they come freighted with problems of many kinds. The question of steroids deserves an entire installment of Doc Gumshoe, so I’ll say no more here. But I will add that both TNFα inhibitors and steroids need to be discontinued well prior to surgery, to minimize the risk of surgical infection.
What about NSAIDs and COX-2 inhibitors?
Surely every Gumshoeland denizen remembers the great controversy about COX-2 inhibitors, in particular Vioxx, yes? In case it has slipped from the forefront of your keen minds, here’s a bit of background.
For a long time, plain old aspirin was the mainstay of management of most aches and pains. Aspirin carries out its analgesic effects by inhibiting the production of prostaglandins, which are messengers of pain, so inhibiting prostaglandin synthesis is evidently a good thing. Except that prostaglandins are also vital in protecting the mucous lining of the GI tract, especially the stomach. Aspirin has, besides its analgesic properties, at least three other properties. It is an antipyretic, meaning that it reduces fever. It is also an anti-inflammatory, and it has antiplatelet (anti-clotting) activity. When the prostaglandin-inhibiting activity of aspirin is combined with its antiplatelet activity, that adds up to a considerably increased risk that people taking high-dose aspirin for long periods will have bleeding in their GI tract that won’t heal because the blood is too slow to clot!
The NSAIDs – non-steroidal anti-inflammatory drugs (the term is an obvious effort to make it clear that these drugs are not steroids and do not have any of the dangerous steroid side effects) also inhibit prostaglandin synthesis via the cyclo-ozygenase (COX) pathway; unlike aspirin, they do not have anti-platelet effects. Therefore patients who take these non-aspirin NSAIDs may be somewhat less affected by GI bleeding, but they can have GI side effects.
The COX pathway has become a source of great interest because of the discovery that there are two of these pathways, labeled COX-1 and COX-2. Most of the NSAIDs – ibuprofen, naprosyn, etc – are non-selective COX inhibitors, meaning that they interact with both pathways. But it turns out that while the COX-1 pathway is the one that produces the beneficial prostaglandins (as well as other necessary GI tract actors), the COX-2 pathway is the one that produces the nasty prostaglandins involved in pain and inflammation. Therefore, agents that selectively inhibited the COX-2 pathway might reduce the bad effects while preserving the useful ones.
This valuable discovery led to the development of the COX-2 inhibitors – Vioxx (rofecoxib) and Celebrex (celecoxib). As nearly everyone on the planet knows, Vioxx hit a pothole very quickly, when it became known that in its clinical trials it had been associated with an unusually high percentage of cardiovascular side effects, including deaths from heart attacks and strokes. Vioxx was taken off the market rather quickly after its introduction. Celebrex was less affected by CV events, and it has remained on the market.
The controversy has not vanished, however. There are indications that all the NSAIDs may, to varying degrees, have CV effects. The risk that an NSAID will contribute to the risk of a CV event is greatest in patients who are already at significant CV risk, such as patients who have already sustained what’s termed an acute coronary syndrome, meaning a heart attack, a stroke, or obstruction of an artery requiring intervention. The NSAID associated with the least CV risk appears to be naproxen (Naprosyn), while the one with the greatest CV risk appears to be diclofenac (Voltaren). The increase in risk in low-risk individuals is very low, difficult to quantify, and causality is difficult to establish. It may be that persons who take NSAIDs chronically and in large doses may be at higher risk for cardiac events due to other causes.
The NSAID dilemma nicely illustrates the calculations that both patients and doctors need to go through in picking a drug. When patients are in pain, quelling the pain is usually their uppermost concern. The choice of a treatment depends largely on the balance between benefit and risk, and it is vital for the patient neither to under- or overestimate the risk.
A very rare side effect that steers millions away from a potentially useful drug
About ten million of us here in the USA and many millions more elsewhere have osteoporosis. As the name suggests, in osteoporosis the bones become porous and crumbly; it is a progressive disease of bone loss. In persons without this condition, bones are constantly being remodeled – in fact, “bone remodeling” is the term for a continuing process whereby old bone tissue is absorbed and replaced with new, stronger bone tissue. The process of building new bone comes to a halt in persons with osteoporosis, and the consequences can be devastating, including spinal and hip fractures that leave many people severely handicapped for life.
A class of drugs called bisphosphonates partly addresses this situation. These drugs, including Fosomax (alendronate), significantly slow the absorption of old bone tissue, but have no effect in terms of building new bone. The preservation of the mature bone greatly reduces the potential for the spinal and hip fractures.
But there is a terrifying side effect that could affect patients taking bisphosphonates – sudden, unexpected shattering of the thigh bone and sometimes erosion of the jawbone. These side effects are extremely rare. The incidence, according to long-term follow up, is something like 0.02%, compared with an incidence of hip fractures in persons with osteoporosis of about 2%. Which is to say that a person with osteoporosis is about one hundred times more likely to experience a hip fracture than the bisphosphonate-associated side effects. Nonetheless, the prospect of those side effects is so terrifying that millions of persons with osteoporosis totally forego bisphosphonate treatment, accepting the prospect of gradually becoming handicapped or even incapacitated, which is about one hundred times greater than the risk of those extremely rare side effects.
However, there are other options for treating osteoporosis that do build new bone, which the bisphosphonates do not do.
To end on a more cheerful note, newer options for treating osteoporosis
One drug that actually fosters new bone growth, already on the market, is Forteo (teriparatide), from Eli Lilly. And a new drug, abaloparatide, so far not on the market, reported highly encouraging Phase III results on August 16th. The drug maker, Radius Health, announced the results of a clinical trial in 2,463 post-menopausal women. Women taking their drug benefited by an 86% reduction in vertebral fractures compared with women taking the placebo, and this result was considered highly statistically significant, with a P value less than 0.001. That P value means that the odds that the results of the trial were due to chance were less than one in a thousand.
Because the mechanisms of action of abaloparatide and teriparatide are totally different from that of the bisphosphonates, it’s thought to be extremely unlikely that these agents would have a similar bone-shattering side effect, but as of this time it’s impossible to rule it out. What these drugs are, essentially, is synthetic analogues of the hormone that foster bone remodeling. They are associated with a range of side effects, but none are in the terrifying class.
It’s not clear that abaloparatide is headed for automatic coronation by the FDA, nor yet that this will result in fame and riches for Radius Health. Predicting the fate of biotech outfits is not Doc Gumshoe’s métier. However, one likely benefit that abaloparatide approval may convey is perhaps driving down the price of Forteo. Eli Lilly, perhaps anticipating that their drug will soon no longer be the only option for actually rebuilding bone in osteoporotic persons, has hugely increased the price of Forteo, by up to 15% every six months. It is now triple what it was in 2010.
And, by the way, the fact that abaloparatide was studied in post-menopausal women certainly does not mean that it would be used in women only. The trial enrolled post-menopausal women because it is this cohort that has the highest likelihood of experiencing osteoporosis. But some men have that disease as well, and the drug, if approved, would be appropriate for men as well.
And finally …
… how should we interpret the information in the package insert?
The package insert/prescribing information/PI should be seen as a sort of triple-function shield. It protects the patient, but it also protects the physician from committing a blunder. And, perhaps most essential, it protects the pharmaceutical company from potentially gigantic legal claims.
The absolutely worst thing a drug company can do is to find bad things in a clinical trial or in post-marketing studies and fail to disclose them to the public. That’s what Merck was accused of in the Vioxx episode, and it has been dreadful in the extreme for the company. Merck has paid out something like $5 billion in settlements, while mostly denying that Vioxx directly caused the large number of deaths in patients taking Vioxx. (One might ask, if not Vioxx, then what killed these people? My answer: remember the data, going back forty years or so, that demonstrated that persons with rheumatoid arthritis had about the same death rate as those with multiple occluded arteries and other severe cardiovascular risk factors. It might just be their underlying diseases that killed them.)
Therefore, the PI includes every conceivable side effect and warning, “failure to warn” being the cardinal sin that condemns pharm companies to purgatory or worse. And, therefore again, the rest of us, folks like you and me, need to read the PI with the tiniest grain of salt. As we saw from the list of Lipitor side effects, in many or even most cases, the frequency of side effects in the patients taking the actual drug is not a whole lot higher than in those taking placebo. And a lot of those side effects are somewhat trivial.
What we need to focus on in the PI are the Warnings and Contraindications sections. We need to sort out the serious and scary side effects from the ones we can fairly easily live with. And, more than anything else, we need to try to balance the risks of taking the drug with the risks of not taking the drug. There’s no sense in risking an episode of torsade de pointes to avoid an episode of hay fever. But it might make sense to accept the risk – about 2 in 10,000 – of a femur shattering, in order to avoid the risk – about 2 in 100 – of a spinal fracture. It’s hard to bring oneself to such calculations, but that’s what it comes down to.
* * * * * *
We’re off on vacation to a small island off the Maine coast for a couple of weeks, no access to email (as well as being off the coast it’s also a bit off the grid), so I won’t be able to respond to comments or questions about this until we get back. But do please keep the comments coming anyway! Best to all, Michael Jorrin (aka Doc Gumshoe)
Dear Doc Gumshoe. Thanks for a great article. I am sure many will be informed and will have had personal experiences with some of the medications you have discussed. I find it very helpful to know the mechanisms that cause drug-drug interactions. I recently noticed the pharmacist had overlooked one, and had my MD change me to a different medication (statin) that did not use the same clearance pathway as another medication I was taking, so I would not loose the effectiveness of either or both. My wife, who has very serious osteoporosis could not tolerate Forteo so we are hoping the Radius drug may work for her. Best, Don
It’s a fascinating domain. I wish I was younger, I would dive into it.
A very nice article. However as one of the rare individuals who has experienced the side effects of Lipitor (all the musculo-skeletal effects that precede rhabdomyolysis); beta blockers (at the lowest dose, I could not walk 50 feet within thirty minutes of taking the medicine, and the effects lasted for hours); and codiene-derived pain medications (vomiting for hours), I can say side effects are no joking matter. Its a good thing that the pharmaceutical companies report these effect, and an equally good thing that my doctors took me off the medications has soon as I reported the side effects.
I am more concerned with the “number needed to treat” (NNT) for a drug like Lipitor, & the fact that the 1 2nd. heart attack “prevented” does not correlate with longer lifespan. the evidence showing only a “benefit” in men under 70 who have had a first heart attack (& only those men [no women], & only under 70). above 70 years old the higher the cholesterol level; the longer the lifespan (in both genders). I want to live longer.
Good morning Doc
Unlike F.E.Smith’s judge, having read your article, I am both wiser & better informed! As someone who has been taking prednisolone (as they call it on this side of the pond) for 40 years I look forward to your next article when you explain in greater detail the less pleasant effects thereof. My own experience of drug side effects reads as follows:-
1. Take prednisolone re pulmonary sarcoidosis.
2. To avoid osteoporosis effects of prednisolne, take alendronic acid tablet.
3. To rectity acid reflux problems caused by taking alendronic acid take H2 antagonist, Ranitidine
I will certainly follow up re Forteo.
On the side effect profile you have to take into consideration the side effects that happened with the placebo. A lot of patients have imagined side effects. It’s when you see a high percentage of effects that there may be a concern and the severity of those effects., on those taking the drug itself.
As a medical doctor, I must say I not only enjoy your writings but also learn/re-learn some of the basics of medical science I had studied years ago in medical school, and some I had not!
Good job, Michael! Have a pleasant and restful vacation.
My best to you,
Charles
I suspect most doctors are not really into the interaction of drugs that a patient might be taking. They are more concern with the drugs that they prescribed. So a useful website can be set up by those who know that warns a patient when they post the various drugs that they are taking and the possible consequence from the unwitting interaction.
Pharmacist should do more then selling drugs. They can warn their customers if they pick up that their customers are consuming various prescribed drugs that could have a bad interaction or long term consequence.
You should choose your pharmacist as you would your physician. My recommendation is to have your prescriptions filled at an independent Pharmacy and develop a relationship with the pharmacist. Not saying chain pharmacies are not good, just harder to develop personal care type relationships. If you have insurance the same copay price at independent pharmacies as chains in almost all instances. Whatever pharmacy you choose you should have ALL your medications filled there, so their scan of your drug profile will pick up any interactions. Ask your pharmacist to do this scan for you.
Speaking of adverse reactions, I have been told that neuropathy, peripheral and other, is ALMOST INEVITABLE after long term use of statins. The same source is selling a therapy regimen employing an “FDA Cleared” Low Level Light Therapy device for the REALLY LOW price of $4995!!! For the money you get the LLLT device and an individualized plan to use it. And, DO NOT be fooled, even though there are a dozen or more such devices “FDA Cleared”, theirs is the ONLY ONE that really works!
I question whether the incidence of Fosamax jaw necrosis is only .02%. In a somewhat small retrospective study published in January, 2009 in the Journal of the ADA, it was 4%. I never trust the results of a single study, be it alarming or comforting.
Of interest to me, is that despite whatever warnings might have been in the PI, the patients got the horrific consequences. If concerned about osteopenia, there are non-drug approaches that I believe do the job.
Very nice article Doc Gumshoe and looking forward to more. Have posted this before and will again for those that have concerns in bone health, http://www.nbihealth.com/default.aspx..I have yet to find any better site that deals with these issues….and backs it up with mounds of clinical data. Well worth the time to take a look.
I strongly believe that all of us approaching “senior” status would benefit from the advice given by Dr Neustadt, There is no denying that broken bones to the senior community has devastating effects on the overall quality of life thereafter and thus becomes a ticking timebomb when it happens.
To BJI’s post previously on LLLT devices, this is something that I have been involved with for several years and still haven’t figured it all out. What I do know is that there is mounting evidence (and probably always has been) on the amazing healing properties of light….but you are right in that it doesn’t have to cost $5K. Both the Chiropractic and Veterinary communities are embracing it for both recovery, pain, etc…but make no mistake, there has to be a financial advantage for most to embrace it….unfortunately the clinical results alone usually is not the driving factor. Let me know if you have the answer.
took prenisone smallest dose my face became atopic of conversation w// a sunburn classifcation also took prodacta on thrd day the blood came from my noze looked like it wudnt stop
lennie519@yahoo.com
I disagree, kind sir, with your article. Please entertain a little logic within the limitations of brevity.
The body, in its natural state, is designed to heal itself. Our challenge lies in providing the tools needed to optimize this remarkable ability.
If a thing happens to be “natural” to the body then chances are the greatest it will help the body. If, on the other hand, a synthetic or foreign substance is introduced, compatibility is defied particularly in the long run.
Current allopathic medicine (Samuel Hahnemann (1755–1843) had it’s original roots in those things natural but strayed far away from that premise long ago. Somehow it all became a profit motive and no longer a “curative.” Just ask yourself: After billions of dollars and likely 100 years of “research,” why do we still have plenty of rising cancer diagnosis? For the short list. [Do check out: The Truth About Cancer 1:58:49 https://www.youtube.com/watch?v=KqJAzQe7_0g ]
Pharmaceutical interests have successfully duped us all and, with that in mind, your article propagates the same erroneous story. Whether you realize it or not. I hope you will consider another way.
I am nothing more than a mom who has successfully raised grown adults, 33 years & running. Starting with a healthy diet (not junk food/chemicals) and reasonable exercise, my family – and numerous others along the way – have so far overcome a multitude of maladies with the administration of natural/homeopathic medicine. No worry with “side affects.”
Honestly have no axe to grind or compensation to receive. I respond in good conscience as a small hope to expand your thinking and finding a better option with far better outcomes for whomsoever may read this.
Be happy!