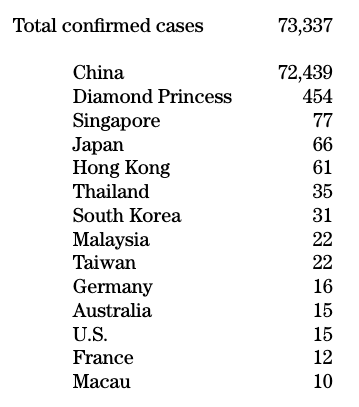
Don’t look to Doc Gumshoe to keep you up to the minute on the spread of the Wuhan coronavirus. The figures I’m quoting below are as of mid-day today, February 18th. These will have changed by the time you see this piece, and they may be unreliable in the first place. But, as of now, here they are:
All others are in the single digits.
The number of deaths as of today was 1,875, all of which were in China except for one each in the U.S., France, Hong Kong, Japan, the Philippines, and Taiwan.
The data above come from the Johns Hopkins coronavirus “dashboard,” which is updated daily. I’ve noticed that the Johns Hopkins data does not always exactly agree with the numbers cited in the news media, which don’t always agree with each other, so there we are.
It’s almost bizarre that the second highest number of cases of the coronavirus were on board the cruise ship Diamond Princess. As of today, the quarantine on the passengers on the Diamond Princess has ended, and many of the passengers are flying home. However, it turns out that 14 passengers had tested positive for the coronavirus, and that despite the assurances that no infected persons would be travelling on those flights, those infected individuals were on those same flights, although they were isolated from the non-infected passengers by means of a large plastic sheet that was hung across the cabin.
In addition to the Diamond Princess, another cruise ship, the Westerdam, carrying more than 1,455 passengers and 802 crew members, was allowed to dock in Cambodia after having been refused permission dock at several other ports. It is unknown how many of those passengers and crew might have been infected with the coronavirus, but since docking, many of those passengers have left Cambodia for points all over the world. Some epidemiologists have raised concerns that the single event of the dispersal of the Westerdam’s passengers to destinations all over the world might be what sets off an uncontrolled global spread of the disease.
As by now you surely know, this novel virus has received an official new name from WHO – COVID 19. WHO does not approve of naming the virus after the place where it first emerged into notoriety. Calling it the Wuhan virus would forever taint the city of Wuhan, says WHO. So the clumsy 2019-nCoV designation is out, and COVID 19 is the name of choice.
What I will have to say about this pandemic – and, yes, it is now officially a pandemic, even though overwhelmingly the majority of cases are in China – is not in the category of breaking news. Instead, I’ll try to focus on the disease itself and how it might be able to be treated.
Potential treatment for COVID 19?
In my previous diatribe, “Warm Bits for a Cold Month,” which posted January 30th, I briefly referred to remdesivir, an antiviral originally developed by Gilead as a treatment for the Ebola virus, but which was not found to be as effective as other options. Remdesivir is also effective against the Marburg virus and has been found to have antiviral activity against such coronaviruses as MERS and SARS.
Precisely one day after that piece posted, on January 31st, The New England Journal of Medicine published an article describing the use of remdesivir in the first US case of what was then called Wuhan coronavirus. (ML Holshue et al, “First Case of 2019 Novel Coronavirus in the United States,” N Engl J Med 2019;1/31)
The patient, a 35-year-old man, had returned to the US on January 15th, after returning from a visit to his family in Wuhan. On January 19th, he presented to an urgent care facility in Snohomish County, Washington, with a four-day history of cough and fever. He reported that he had seen a health alert from the Centers for Disease Control and Prevention (CDC) about this novel coronavirus outbreak in China and decided to seek medical attention.
The urgent care clinician reported the case to the Washington Department of Health, which in turn notified the CDC Emergency Operations Center. The patient was tested for the Wuhan coronavirus, and the next day, January 20th, the report came back that the swab samples had tested positive for that virus. Those specimens as well as stool samples indicated a high viral load.
For the next four days, the patient remained relatively stable, although with a non-productive cough, intermittent fevers, and periods of abnormally fast heart rates. However, on hospital day five, radiographic findings showed evidence of pneumonia in the lower left lung. He was subsequently started on supplemental oxygen, plus antibiotics to prevent hospital-acquired pneumonia. On hospital day six, X-rays showed that the pneumonia had spread to the right lung as well.
In response to worsening symptoms which were consistent with reports of the symptoms of what was then called 2019-nCoV, the clinicians decided to initiate treatment with intravenous remdesivir.
On hospital day eight, after one full day of treatment with remdesivir, the patient’s clinical condition improved. It was possible to discontinue supplemental oxygen, and the patient’s oxygen saturation, while breathing ambient air, returned to normal levels.
As of January 30th, the patient remained hospitalized, but his body temperature was normal and all symptoms had resolved with the exception of his cough, which was declining in severity
It is by no means certain that the Snohomish County patient’s remission was due to remdesivir, but based on that experience, a couple of clinical trials of remdesivir are now starting up in China. A hospital in Wuhan plans to randomize 270 patients with mild-to-moderate pneumonia due to the Wuhan virus to receive either remdesivir or placebo. And in another trial, remdesivir with be tested in two patient cohorts – one with mild-to-moderate pneumonia and the other with more serious cases. The plan is to enroll a total of 761 subjects.
Gilead provided the remdesivir for the Snohomish County patient and is providing the drug for the two clinical trials in China.
Meanwhile, Chinese pharmaceutical companies have been keeping their eyes open. No sooner had the news of remdesivir become public than a Chinese pharma, BrightGene, was on it. A few days ago, BrightGene announced to the financial community that it had successfully copied remdesivir’s active ingredient and had begun to produce it on a large scale. Their stock jumped 20% on the news, which is apparently the maximum price move that market permits.
BrightGene does not plan to get into a patent war with Gilead. The final marketing of their generic version, which is still in development, will require permission from Gilead, which holds the original patent on remdesivir. On its part, Gilead has said that it does not plan to oppose BrightGene’s marketing of a generic version of remdesivir.
"reveal" emails? If not,
just click here...
Researchers at the Chinese Academy of Sciences’ Wuhan Institute of Virology also announced that they had applied for a patent on the use of remdesivir to treat the novel coronavirus.
Gilead’s CEO Daniel O’Day recently emphasized that Gilead owns all patents around remdesivir including for its use in treating coronaviruses. However, he said that the top priority is to evaluate remdesivir in well-conducted clinical trials and only then, if its efficacy is confirmed, ramp up production of the drug. Gilead is not planning to get into patent disputes.
The whole issue around the Chinese copy of remdesivir brings up the highly contentious matter of Chinese theft of US and other intellectual property. This theft, or appropriation, is often justified as a matter of humanitarian necessity. The position is that the needs of a large population threatened by a potentially deadly disease trump the profit motives of an already rich American pharmaceutical company.
That’s as may be, but Doc Gumshoe is not aware of any Chinese pharmaceutical outfits sacrificing their profits in the name of humanitarian need.
Another HIV drug, Kaletra, from AbbVie, is also being investigated as a possible antagonist to COVID 19. As the world’s infectious disease specialists search for drugs that might be used to treat victims of COVID 19, they are first looking at existing antivirals to see if any of them might work. They will go about this search with the utmost diligence, but they are seriously hampered by the circumstances of this outbreak. The immense majority of the persons infected with this virus are in China, and are thus not available for clinicians in the rest of the world to test these antivirals. And as yet there are no laboratory animals infected with COVID 19. The bulk of the research will have to be carried out in China.
While the attention of the medical community has been focused on finding a treatment that specifically targets the evil virus, patients have routinely been treated with supportive care, including ondasetron for the nausea associated with the acute respiratory symptoms, guaifenesin for congestion, acetaminophen and ibuprofen as analgesics, and vancomycin to prevent secondary hospital-acquired infections.
A role for Chinese traditional medicine
In the meantime, while Chinese physicians trained in Western medicine are trying to treat victims of COVID 19 with a cocktail of antivirals that have demonstrated some effectiveness in HIV, Chinese traditional medicine proposes its own cocktails. One is named the Peaceful Palace Bovine Pill, and it is being recommended by the Chinese government, right up at the highest levels. President Xi Jinping has stated that officials should place as much importance of traditional Chinese medicine as they do on Western medicine. His government has called on these remedies to be promoted in China’s “Belt and Road” trade route, which is China’s push to build infrastructure in other parts of the world.
The Peaceful Palace Bovine Pill, however, is made with cattle gallstones, buffalo horn, jasmine, and pearl. It has been pointed out that there is no evidence that the Peaceful Palace Bovine Pill and other remedies such as the roots of various plants and licorice can have any effectiveness against this virus. Practitioners of Chinese traditional medicine say that it may help ease some symptoms such as swelling of the lungs. A professor of pharmacology at Yale University School of Medicine, Cheng Yung-Chi, asserts that using Chinese traditional medicine is the correct approach and should be given the benefit of the doubt. The evidence will emerge, he says.
A manufacturer of traditional Chinese medicines in Hubei (the province where Wuhan is located) has reported that they are turning out more than 20,000 prescriptions of the Peaceful Palace Bovine Pill per day, at the request of the Chinese government.
Another traditional remedy called Shuanghuanglian, which uses flowers like honeysuckle and forsythia, was recommended by two government-backed research institutions which published a study saying that it inhibited the growth of the novel coronavirus. In many Chinese cities, people stood in crowded lines for hours to buy this medicine. Chen Xi, a professor at the Yale School of Public Health commented that it was a better idea to avoid those crowds than to buy a medicine for which there was no evidence. It might inhibit the growth of the virus in a test tube, but that is not equivalent to effective treatment.
It has also been emphasized by some observers that accurate dosing is nonexistent in Chinese traditional medicine. Nobody has tried to establish the right dose, and the traditional medicines are not sold in precise doses. It would be as easy to underdose as to overdose, even assuming that there is a correct dose.
And it has also been noted that in one trial, dozens of women who were taking Chinese herbs as part of a weight loss program ended up with kidney failure.
I should point out that at this moment, the evidence supporting the efficacy of remdesivir is not much different from the evidence supporting the efficacy of the Peaceful Palace Bovine Pill. Remdesivir has been used in one patient, whose symptoms improved markedly after just one day, but there is no evidence that it was the remdesivir that caused the improvement. The patient in question may have recovered spontaneously. His own immune system may have rallied and defeated the virus on its own, and remdesivir may have had nothing to do with his recovery.
As for the Peaceful Palace Bovine Pill, there is neither clinical evidence that the components of that combination actually have any effect on the virus itself, nor yet statistically valid evidence that it works better than placebo, nor yet any indication of what its mechanism of action might be. The evidence supporting that medicine, as well as many Chinese traditional medicines, is that some patients’ symptoms improve after being given those medicines. That’s what we call the post hoc propter hoc fallacy – if event B happens after event A, then event A must be the cause.
The fundamental difference is that remdesivir will be tested in rigorous clinical trials – those two aforementioned trials in China, for a start, and then certainly more trials in other parts of the world.
Might a vaccine against COVID 19 be in the works?
The answer appears to be yes. The National Institutes of Health is working with Moderna Therapeutics to develop a vaccine against COVID 19. However, it won’t be so easy to get it into wide use. Developing a candidate vaccine that might work to reduce the incidence of an infection like COVID 19 might be the easy part. Researchers would likely be guided by the immune responses of persons who have had the infection and survived it. But then the candidate vaccine would have to be tested very scrupulously to verify that vaccinated persons, when exposed to the virus, would resist infection. In vitro studies and studies in laboratory animals would go a certain way towards providing the necessary evidence, but before such a vaccine could be recommended to the population at large, some human testing would be needed.
Other players in the effort to create a vaccine against COVID 19 are Johnson & Johnson and the U. S. Biomedical Advanced Research and Development Authority (BARDA). J & J has considerable experience in providing antivirals for use in China, and described a multipronged approach, employing its proprietary technologies AdVac and PER.C6 to the development of this vaccine. J & J is pooling resources with BARDA to accelerate the progress of a vaccine candidate into development and testing. The project will be co-funded by J & J’s Janssen and BARDA.
Another pharmaceutical outfit that has jumped into the race to develop a COVID 19 vaccine is Sanofi, which is joining with the BARDA unit to develop a recombinant DNA vaccine based on prior research conducted by Protein Sciences, a flu vaccine biotech acquired by Sanofi in 2017.
The next step would be mass-producing the vaccine, which is a complex process – in a sense, the processes that our own organisms go through in developing immunity must be duplicated in the manufacturing facility.
Children and COVID 19
It appears that young children are by and large escaping the coronavirus. The median age of the patients who have been diagnosed as being infected with the virus is between 49 and 56 years, and, as expected, it is the older patients, especially those with other disorders, who have the highest fatality rate. And as increasing numbers are being infected, the median age of those infected has kept skewing older.
It’s by no means obvious why this should be the case. It’s not that children are not being exposed to this highly infectious coronavirus. Along with their older family members, children in the Hubei province are prevented from leaving the area where the transmission of the virus is highest.
An explanation that has been put forward by infectious disease specialists is that children might be protected by the innate immune response, which is passed from mother to child. This is consistent with observations that, although children are certainly susceptible to many infectious diseases such as chickenpox and measles, these infections have far more severe consequences when they occur in adults. It has been observed that children are also less likely to be infected by other coronavirus diseases such as SARS and MERS.
The innate immune response is an early response that confers protection against broad groups of pathogens. In contrast, the acquired immune response is developed in the individual person when a specific invader is recognized and identified by the immune system as a hostile invader. Once a person’s immune system has spotted a specific pathogen and mounted a response to it, a degree of immunity against that pathogen persists. Even though the pathogen – for example, the influenza virus – may mutate, a certain degree of immunity against the mutated virus is apt to persist. That’s why, even if the current flu vaccine in an flu season may not exactly match the flu in circulation in a particular season, some immunity persists, and episodes of the flu are apt to be less severe.
It is possible that the innate immune response in children in that region of China may have developed in response to previous exposure to other coronaviruses. This suggests that, despite the alarming rapidity that the novel coronavirus has shown in infecting the many tens of thousands in that province, a degree of herd immunity may exist there, whose effect is not preventing infection, but mitigating the effects of the disease.
Please note that I put that forward as my own speculation, and not anyone’s expert opinion. Supporting that speculation is the fairly low fatality rate from COVID 19 – somewhere in the neighborhood of 2.5%. This is not nothing, but it’s far from the Black Death.
… and how is China addressing the treatment of its infected multitude?
From what I have been able to glean from the avalanche of news about the outbreak, treatment seems to consist in locking the patients up, whether in their homes or in improvised shelters. Yes, they’re working on a drug or perhaps more than one. But many infected people can’t get to a hospital – the hospitals have no room, and the Chinese medical system appears to be entirely overwhelmed. The main objective of the official Chinese response is to prevent, as much as possible, the disease from spreading to other parts of China. This makes sense from a national perspective, but confining huge numbers of people, infected and not infected, almost seems to be an abandonment of the population of the vulnerable. It has been reported that somewhere around 700 million people in China are, in one way or another, being isolated and contained. It seems to me that what they’re saying is, “We’ll do what little we can for the population of Wuhan and its environs, but let’s keep the rest of China safe.”
As to the likelihood of the spread of COVID 19 in developed parts of the world, I would say the odds of any one of us escaping this disease are pretty good. So far, as near as I can tell, there are 15 infected people in the US and one has perished. There has been an outbreak at a ski resort in the Swiss Alps, brought by one person coming from China. There have been no cases in Indonesia – a near neighbor. The dispersion of the 1,455 passengers from the Westerdam, many of whom were at least exposed to COVID 19, presents the greatest threat. The great fears of epidemiologists are that there would be outbreaks in crowded countries with limited medical facilities, such as in sub-Saharan Africa. We can hope that it doesn’t happen, and in the meantime keep on being careful.
* * * * * * *
My intention, when I put my fingers to the keyboard, was to cover a bunch of current news of interest in this piece before progressing to a promised piece about Parkinson’s disease and Parkinsonism. But my inbox got swamped daily with stuff about this coronavirus, a lot of which didn’t seem to make it into the regular news, so I gave that subject a priority designation.
Keep up the comments, and best to all, Michael Jorrin (aka Doc Gumshoe).
[ed. note: Michael Jorrin, who I dubbed “Doc Gumshoe” many years ago, is a longtime medical writer (not a doctor). He shares his thoughts on health and medicine topics with our readers a couple times a month (you can see his past columns here), and while he does not focus on the investment prospects of any idea or company he has agreed to our trading restrictions]
I surprised that the Chinese government didn’t mention Chinese star anise. Its most effective antiviral ingredient is the basis for Tamiflu. Personally I prefer the synergy of the whole herb or a somewhat concentrated extract.
Virus spread by slops at market place as they have been for 2500+ years of humanity
The Chinese just trucked 50 tons of vitamin C (inexpensive ascorbic acid) from a different Province… to solve their problem… Are they just stupid? Or crazy? Or crazy like a fox? Or putting their money, not where their mouth is; but, where it will work… That way they can laugh at US all the way to the bank… & they are smart enough to own the bank… We are not…
They’re also sure enough that rhino horn is an aphrodisiac to effectively green-light the extinction of rhinos. All cultures have blind spots, and are absolutely certain of things for which the scientific evidence (or even common sense rationale) is completely lacking in other cultures.
You have obviously not read the science (none of which comes from China) from the U.S. NIH , Mayo clinic , ( scince there are literaly thousands of double blind studies I haven’t the time or inclination to list…) Do not listen to opinion; including my opinion, It is for sale for the right price … I haven’t the time to wade through opinion… Data is shorter & is not for sale, as long as we always remember that figures don’t lie but liars figure… That is why only the numbers count , not abstracts, titles, or digests… Life & death, It’s your choice; no M.D. is going to take our place in the coffin… I would guess there are some very smart chinese scientists… why else would we have them make our medicines & vaccines in China??? No worries the stupid make themselves extinct…
Peoples Liberation Army has to do something, that is the social contract governments have with population- no down side to Vitamin C plus they probably could get there hands on a bunch. Virus has already mutated at least once. I can only imagine the devastation coming in countries like India and Mexico were health care was already non-existent for majority of population and cities are overcrowded.
Great and informative article from a medical prospective. What stocks do u all think will rally, even if temporarily, during this outbreak?
In 1979 I began college for a pharmacy degree. My father in law was an emeritus professor at University of Michigan school of Public Health. He was also a member of the World Health Organization. He was a well known scientist with military background in biochemical warfare. A major in US Army during WWII. He spoke of the “bad” things the WHO scientists were doing. One I have never forgotten was the “contest” in Africa and other countries to produce a new, unique organism. This story meant nothing to me until sometime later as an intern I was providing meds for AIDS patients at Veterans Hospital. I did research and found the same story my father in law told me with details. Story debunked involving WHO but said dna from bovine and a monkey was used to make a new “organism”. Test subjects were injected with new organism. Investigation found no documentation of study. Never underestimate nor totally disregard information things never are as they may seem! I am 76 years old—eventually the truth will be known and there will be no accountability. Same with Coronavirus.
cmilone, Unfortunately and as my interest was just starting to peak, your missive got truncated. Is there a way you can continue your narrative, maybe by breaking it up into a few lines at a time until the Gumshoe system is fixed?
Doc,
You might find the following interesting about statistical numbers for Covid-19 from Dr. Frank.
https://dispensationalpublishing.com/blog/
7.6 Billion world population, 43 million births/yr. avg., 18 million deaths/yr. avg. Wuhun Virus deaths 184,000 to date. 0.1% of 10 million population.
https://wattsupwiththat.com/daily-coronavirus-covid-19-data-graph-page/
Self explanatory counter opinion https://principia-scientific.org/aids-research-pioneer-blows-whistle-on-dr-fauci/#comments